MTOR (FK506 binding protein 12-rapamycin associated protein 1)
2008-06-01 Deborah A Altomare , Joseph R Testa AffiliationFox Chase Cancer Center, 333 Cottman Avenue, Philadelphia, PA 19111, USA
Identity
HGNC
LOCATION
1p36.22
IMAGE

LOCUSID
ALIAS
FRAP,FRAP1,FRAP2,RAFT1,RAPT1,SKS
FUSION GENES
DNA/RNA
Note
A map of the genomic organization of the human FRAP1 gene can be found at http://www.ncbi.nlm.nih.gov/projects/sviewer/?id=NC_000001.9&v=11081381..11252951
Description
The FRAP1 gene encompasses approximatively 156 kb and contains 58 exons. The gene resides on the minus strand. Reported location on human chromosome 1 is between 11,089,179-11,245,151 bases in NCBI36 coordinates and 11,089,180-11,245,176 bases in ensemble49 coordinates.
Transcription
Transcript length is 8,680 bp.
Pseudogene
No human pseudogene known.
Proteins
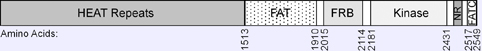
The amino acid residues corresponding to the FRAP1 (alias mTOR) protein domains are reported in pfam (see below: external links)
Pfam: PF00454: Phosphatidylinositol 3- and 4-kinase (2181-2431)
Pfam: PF02259: FAT domain (1513-1910)
Pfam: PF02260: FATC domain (2517-2549)
Pfam: PF08771: FKBP12 Rapamycin Binding domain (2015-2114)
Description
The amino terminus of FRAP (alias, mTOR) consists of several tandem HEAT ( Huntingtin, EF2, A subunit of PP2A, TOR1) repeats) that are implicated in protein-protein interations (Hay and Sonenberg, 2004; Bhaskar and Hay, 2007). Each HEAT repeat contains two alpha helices of approximatively 40 amino acids.
The carboxy-terminal half contains two FAT (FRAP, ATM, TRAP) domains.
Upstream of the catalytic domain is the FRB (FKBP12-rapamycin binding) domain.
The catalytic domain has sequence similarity to the catalytic domain of phosphatidylinositol kinase (PIK), which is homologous to a family of other protein kinases termed PIKK (PIK-related kinase).
mTOR also contains a putative negative regulatory (NR) domain between the catalytic domain and FATC.
The FATC (FRAP, ATM, TRRAP C-terminal) domain is essential for the kinase activity. The FATC and FAT domains are thought to interact in a way the exposes the catalytic domain.
The protein consists of 2549 amino acids, with a predicted molecular weight of 288,891 Da.
The ternary complex of human FK506-binding protein ( FKBP12), the inhibitor rapamycin, and the FKBP12-rapamycin-binding (FRB) domain of human FRAP has been crystallized at a resolution of 2.7 angstroms (Choi et al., 1996), and then refined at 2.2 angstroms (Liang et al., 1999).
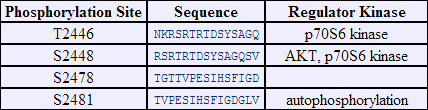
Phosphorylation sites of FRAP (alias, mTOR) are reported in http://www.phosphosite.org .
The carboxy-terminal half contains two FAT (FRAP, ATM, TRAP) domains.
Upstream of the catalytic domain is the FRB (FKBP12-rapamycin binding) domain.
The catalytic domain has sequence similarity to the catalytic domain of phosphatidylinositol kinase (PIK), which is homologous to a family of other protein kinases termed PIKK (PIK-related kinase).
mTOR also contains a putative negative regulatory (NR) domain between the catalytic domain and FATC.
The FATC (FRAP, ATM, TRRAP C-terminal) domain is essential for the kinase activity. The FATC and FAT domains are thought to interact in a way the exposes the catalytic domain.
The protein consists of 2549 amino acids, with a predicted molecular weight of 288,891 Da.
The ternary complex of human FK506-binding protein ( FKBP12), the inhibitor rapamycin, and the FKBP12-rapamycin-binding (FRB) domain of human FRAP has been crystallized at a resolution of 2.7 angstroms (Choi et al., 1996), and then refined at 2.2 angstroms (Liang et al., 1999).
Phosphorylation sites of FRAP (alias, mTOR) are reported in http://www.phosphosite.org .
Expression
Expressed is found in numerous tissues, with high levels in testis.
Localisation
Localization is predominantly cytoplasmic, but the protein is also associated with mitochondrial, endoplasmic reticulum and Golgi membranes (Guertin and Sabatini, 2007). A fraction of protein also may shuttle between the nucleus and cytoplasm.
Function
There are more than 2500 articles specifically referring to FRAP1 or mTOR in PubMed. mTOR is central to several key cellular pathways including insulin signaling, regulation of eIF4e and p70S6 kinase, and hypoxia induced factor 1alpha ( HIF1alpha ) stimulation of vascular endothelial growth factor ( VEGF ). These pathways affect several processes including cell growth (size), protein translation, ribosome biogenesis, regulation of cell cycle progression, response to nutrients and cellular stress, angiogenesis, cell polarity and cytoskeletal reorganization. mTOR also has been shown to play a role in the regulation of autophagy (Pattingre et al., 2008), an adaptive cellular response to nutrient starvation whereby a cytoplasmic vacuole or autophagosome engulfs cellular macromolecules and organelles for degradation.
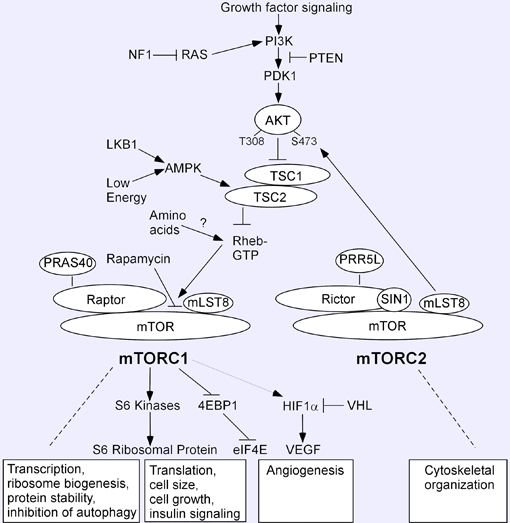
mTOR protein exists in two functionally distinct complexes named mTOR complex 1 (mTORC1) and complex 2 (mTORC2) (see figure under "Implicated in"). The regulator of mTORC1 signaling and kinase activity is the ras-like small GTPase Rheb (Ras homologue enriched in brain), which binds directly to the mTOR catalytic domain and enables mTORC1 to attain an active configuration (Avruch et al., 2006). Insulin/IGF stimulates the accumulation of Rheb-GTP through activated AKT and subsequent inhibition of the Rheb-GTPase-activating function of the tuberous sclerosis ( TSC1 / TSC2 ) heterodimer. Energy depletion decreases Rheb-GTP through the action of adenosine monophosphate-activated protein kinase (AMPK) to phosphorylate TSC2 and stimulate its Rheb-GTPase activating function and also HIFalpha-mediated transcriptional responses that act upstream of the TSC1/2 complex. Amino-acid depletion inhibits mTORC1 by acting predominantly downstream of the TSC complex, by interfering with the ability of Rheb to bind to mTOR.
As shown below, mTORC1 contains the core components mTOR, Raptor (regulatory associated protein of mTOR), and mLST8/GbetaL (G protein beta-subunit-like protein). It is the mTORC1 complex that is characteristically sensitive to inhibition by rapamycin. mTORC1 is a major regulator of ribosomal biogenesis and protein synthesis, largely through the phosphorylation/inactivation of the 4E-BPs (4E-binding proteins) and the phosphorylation/activation of S6K (ribosomal S6 kinase). The binding of S6K1 and 4E-BP1 to raptor requires a TOR signaling (TOS) motif, which contains an essential phenylalanine followed by four alternating acidic and small hydrophobic amino acids (Schalm and Blenis, 2002). Recently, a TOS motif also has been identified in the N terminus of HIF1alpha, which has been shown to interact with Raptor (Land and Tee, 2007). Furthermore, activation of mTOR by Rheb overexpression enhanced HIF1alpha activity and VEGF-A secretion under hypoxic conditions, whereas the mTOR inhibitor rapamycin blocked the pathway.
PRAS40 (proline-rich AKT substrate 40 kDa) is a novel mTOR binding partner that mediates AKT signals to mTOR independently of TSC1/TSC2 (Vander Haar et al., 2007). Hence, PRAS40 and Rheb are postulated to co-regulate mTORC1 (Guertin and Sabatini, 2007). PRAS40 binds mTORC1 via Raptor, and is an mTOR phosphorylation substrate (Thedieck et al., 2007). Moreover, PRAS40 binds the mTOR kinase domain and its interaction with mTOR is induced under conditions that inhibit mTOR signaling, such as nutrient or serum deprivation or mitochondrial metabolic inhibition (Vander Haar et al., 2007). PRAS40 contains a variant TOS motif and competes with S6K1 and 4E-BP1 by functioning as a direct inhibitor of substrate binding (Oshiro et al., 2007, Wang et al., 2007).
mTORC2 contains mTOR, Rictor (rapamycin-insensitive companion of mTOR), SIN1 (SAPK interacting protein) and mLST8/GbetaL. Proline rich protein 5-like (PRR5) protein also binds specifically to mTORC2, via Rictor and/or SIN1 (Thedieck et al., 2007). mTORC2 has been shown to regulate cell-cycle-dependent polarization of the actin cytoskeleton. Although not as sensitive to rapamycin as mTOR1, mTORC2 may be affected by prolonged rapamycin exposure in some cell types (Sarbassov et al., 2006; Zeng et al., 2007). However, the regulation of mTORC2 is largely unknown and does not function downstream of Rheb (Blaskar and Hay, 2007).
Direct genetic evidence for the importance of various components of the mTORC1 and/or mTORC2 complexes was provided by targeted disruption studies in mice (Guertin et al., 2006). Mice null for mTOR, as well as those lacking Raptor die early in embryonic development. However, mLST8-null embryos survive until e10.5 and resemble embryos missing Rictor. Collectively, mTORC1 function was found to be essential in early development, mLST8 was required only for mTORC2 signaling, and mTORC2 was found to be a necessary component of the AKT-FOXO and PKCalphaalpha pathways.
mTOR protein exists in two functionally distinct complexes named mTOR complex 1 (mTORC1) and complex 2 (mTORC2) (see figure under "Implicated in"). The regulator of mTORC1 signaling and kinase activity is the ras-like small GTPase Rheb (Ras homologue enriched in brain), which binds directly to the mTOR catalytic domain and enables mTORC1 to attain an active configuration (Avruch et al., 2006). Insulin/IGF stimulates the accumulation of Rheb-GTP through activated AKT and subsequent inhibition of the Rheb-GTPase-activating function of the tuberous sclerosis ( TSC1 / TSC2 ) heterodimer. Energy depletion decreases Rheb-GTP through the action of adenosine monophosphate-activated protein kinase (AMPK) to phosphorylate TSC2 and stimulate its Rheb-GTPase activating function and also HIFalpha-mediated transcriptional responses that act upstream of the TSC1/2 complex. Amino-acid depletion inhibits mTORC1 by acting predominantly downstream of the TSC complex, by interfering with the ability of Rheb to bind to mTOR.
As shown below, mTORC1 contains the core components mTOR, Raptor (regulatory associated protein of mTOR), and mLST8/GbetaL (G protein beta-subunit-like protein). It is the mTORC1 complex that is characteristically sensitive to inhibition by rapamycin. mTORC1 is a major regulator of ribosomal biogenesis and protein synthesis, largely through the phosphorylation/inactivation of the 4E-BPs (4E-binding proteins) and the phosphorylation/activation of S6K (ribosomal S6 kinase). The binding of S6K1 and 4E-BP1 to raptor requires a TOR signaling (TOS) motif, which contains an essential phenylalanine followed by four alternating acidic and small hydrophobic amino acids (Schalm and Blenis, 2002). Recently, a TOS motif also has been identified in the N terminus of HIF1alpha, which has been shown to interact with Raptor (Land and Tee, 2007). Furthermore, activation of mTOR by Rheb overexpression enhanced HIF1alpha activity and VEGF-A secretion under hypoxic conditions, whereas the mTOR inhibitor rapamycin blocked the pathway.
PRAS40 (proline-rich AKT substrate 40 kDa) is a novel mTOR binding partner that mediates AKT signals to mTOR independently of TSC1/TSC2 (Vander Haar et al., 2007). Hence, PRAS40 and Rheb are postulated to co-regulate mTORC1 (Guertin and Sabatini, 2007). PRAS40 binds mTORC1 via Raptor, and is an mTOR phosphorylation substrate (Thedieck et al., 2007). Moreover, PRAS40 binds the mTOR kinase domain and its interaction with mTOR is induced under conditions that inhibit mTOR signaling, such as nutrient or serum deprivation or mitochondrial metabolic inhibition (Vander Haar et al., 2007). PRAS40 contains a variant TOS motif and competes with S6K1 and 4E-BP1 by functioning as a direct inhibitor of substrate binding (Oshiro et al., 2007, Wang et al., 2007).
mTORC2 contains mTOR, Rictor (rapamycin-insensitive companion of mTOR), SIN1 (SAPK interacting protein) and mLST8/GbetaL. Proline rich protein 5-like (PRR5) protein also binds specifically to mTORC2, via Rictor and/or SIN1 (Thedieck et al., 2007). mTORC2 has been shown to regulate cell-cycle-dependent polarization of the actin cytoskeleton. Although not as sensitive to rapamycin as mTOR1, mTORC2 may be affected by prolonged rapamycin exposure in some cell types (Sarbassov et al., 2006; Zeng et al., 2007). However, the regulation of mTORC2 is largely unknown and does not function downstream of Rheb (Blaskar and Hay, 2007).
Direct genetic evidence for the importance of various components of the mTORC1 and/or mTORC2 complexes was provided by targeted disruption studies in mice (Guertin et al., 2006). Mice null for mTOR, as well as those lacking Raptor die early in embryonic development. However, mLST8-null embryos survive until e10.5 and resemble embryos missing Rictor. Collectively, mTORC1 function was found to be essential in early development, mLST8 was required only for mTORC2 signaling, and mTORC2 was found to be a necessary component of the AKT-FOXO and PKCalphaalpha pathways.
Homology
mouse (Mus musculus): Frap1, 99 % Amino Acid Similarity with Human FRAP1
rat (Rattus norvegicus): Frap1, 99% Amino Acid Similarity with Human FRAP1
dog (Canis familiaris): FRAP1, 99% Amino Acid Similarity with Human FRAP1
worm (Caenorhabditis elegans): B0261.2b, 51% Amino Acid Similarity with Human FRAP1
fruit fly (Drosophila melanogaster): Tor, 64% Amino Acid Similarity with Human FRAP1
rat (Rattus norvegicus): Frap1, 99% Amino Acid Similarity with Human FRAP1
dog (Canis familiaris): FRAP1, 99% Amino Acid Similarity with Human FRAP1
worm (Caenorhabditis elegans): B0261.2b, 51% Amino Acid Similarity with Human FRAP1
fruit fly (Drosophila melanogaster): Tor, 64% Amino Acid Similarity with Human FRAP1
Mutations
Note
No mutations are reported to date.
Implicated in
Entity name
Various cancers and hamartoma syndromes
Note
Activation of mTOR signaling is associated with several hamartoma syndromes, as well as in cancer.
Disease
Among the dominantly inherited disorders classified as phakomatoses are tuberous sclerosis 1 and 2, Peutz-Jeghers syndrome, Cowden disease, neurofibromatosis 1 and neurofibromatosis 2, and von-Hippel-Lindau disease (Tucker et al., 2000). These disorders are characterized by scattered hamartomatous or adenomatous "two-hit" lesions that have a low probability of becoming malignant. These particular disorders are caused by germline mutations of certain tumor suppressor genes, i.e., TSC2 / TSC1, LKB1, PTEN, NF1 / NF2 and VHL, respectively, encoding proteins that intersect with the AKT/mTOR signaling pathway (Altomare and Testa, 2005).
Germline mutations of TSC1 and TSC2 each give rise to the hereditary disorder known as tuberous sclerosis complex (TSC) (Astrinidis and Henske, 2005; Jozwiak et al., 2008). Hamartomas arise in the central nervous system, kidney, heart, lung, and skin, with occasional tumors progressing to malignancy (i.e., renal cell carcinoma). In TSC tumor cells, biallelic inactivation of TSC2 or TSC1 results in constitutive mTOR activity, independent of AKT activation status. Aside from TSC, another rare lung disease known as pulmonary lymphangioleiomyomatosis (LAM) occurs from somatic or genetic mutations of TSC1 or TSC2 that lead to the activation of downstream mTOR (Krymskaya, 2008). These findings have provided rationale for the first rapamycin clinical trial for LAM (Goncharova and Krymskaya, 2008).
The LKB1 tumor suppressor/AMPK pathway is an alternate means of inactivating TSC2 and contributing to constitutive mTOR activation (Inoki et al., 2005; Kwiatkowski and Manning, 2005). The kinase controlling AMPK (AMP-activated protein kinase) has been identified as LKB1, which is encoded by the gene inactivated in Peutz-Jeghers syndrome, a disorder characterized by multiple gastrointestinal hamartomatous polyps. There is now experimental evidence that Peutz-Jeghers polyposis could be suppressed by targeting mTOR (Wei et al., 2008).
Germline PTEN mutations occur in 80% of patients with Cowden disease, a heritable multiple hamartoma syndrome with a high risk of breast, thyroid and endometrial carcinomas (Gustafson, et al., 2007). Decreased or absent expression of PTEN results in constitutive activation of the AKT/mTOR pathway.
Loss-of-function mutations in NF1 contributes to the neurofibromatosis type I familial cancer syndrome, which is characterized by benign neurofibromas and occasional malignant peripheral nerve sheath tumors (MPNSTs), as well as hamartomatous lesions of the eye, myeloid malignancies, gliomas, and pheochromocytomas. The NF1-encoded protein, neurofibromin, functions as a Ras-GAP, and deregulation of Ras due to NF1 inactivation is postulated to contribute to tumor development. Activated Ras signaling to PI3K results in activation of the AKT/mTOR pathway (Johannessen et al., 2005). The mTOR inhibitor rapamycin has been shown to suppress the growth of NF1-associated malignancies in a genetically modified mouse model (Johannessen et al., 2008). Like NF1, NF2 also can regulate AKT/mTOR signaling (Scoles, 2008); the NF2-encoded protein, merlin, does so by binding to PIKE (phosphatidylinositol 3-kinase enhancer).
Germline inactivation of the von Hippel-Lindau tumor suppressor gene (VHL) causes hamartomatous tumors associated with the von-Hippel-Lindau syndrome. Moreover, most renal cell carcinomas have biallelic alterations in the von VHL gene, resulting in the accumulation of hypoxia-inducible factors 1 and 2 , and downstream targets including vascular endothelial growth factor (VEGF) (Cho et al., 2007). The observed clinical efficacy of mTOR inhibitors in patients with renal cell carcinoma may be mediated in part by the dependence of efficient hypoxia-inducible factor translation.
Germline mutations of TSC1 and TSC2 each give rise to the hereditary disorder known as tuberous sclerosis complex (TSC) (Astrinidis and Henske, 2005; Jozwiak et al., 2008). Hamartomas arise in the central nervous system, kidney, heart, lung, and skin, with occasional tumors progressing to malignancy (i.e., renal cell carcinoma). In TSC tumor cells, biallelic inactivation of TSC2 or TSC1 results in constitutive mTOR activity, independent of AKT activation status. Aside from TSC, another rare lung disease known as pulmonary lymphangioleiomyomatosis (LAM) occurs from somatic or genetic mutations of TSC1 or TSC2 that lead to the activation of downstream mTOR (Krymskaya, 2008). These findings have provided rationale for the first rapamycin clinical trial for LAM (Goncharova and Krymskaya, 2008).
The LKB1 tumor suppressor/AMPK pathway is an alternate means of inactivating TSC2 and contributing to constitutive mTOR activation (Inoki et al., 2005; Kwiatkowski and Manning, 2005). The kinase controlling AMPK (AMP-activated protein kinase) has been identified as LKB1, which is encoded by the gene inactivated in Peutz-Jeghers syndrome, a disorder characterized by multiple gastrointestinal hamartomatous polyps. There is now experimental evidence that Peutz-Jeghers polyposis could be suppressed by targeting mTOR (Wei et al., 2008).
Germline PTEN mutations occur in 80% of patients with Cowden disease, a heritable multiple hamartoma syndrome with a high risk of breast, thyroid and endometrial carcinomas (Gustafson, et al., 2007). Decreased or absent expression of PTEN results in constitutive activation of the AKT/mTOR pathway.
Loss-of-function mutations in NF1 contributes to the neurofibromatosis type I familial cancer syndrome, which is characterized by benign neurofibromas and occasional malignant peripheral nerve sheath tumors (MPNSTs), as well as hamartomatous lesions of the eye, myeloid malignancies, gliomas, and pheochromocytomas. The NF1-encoded protein, neurofibromin, functions as a Ras-GAP, and deregulation of Ras due to NF1 inactivation is postulated to contribute to tumor development. Activated Ras signaling to PI3K results in activation of the AKT/mTOR pathway (Johannessen et al., 2005). The mTOR inhibitor rapamycin has been shown to suppress the growth of NF1-associated malignancies in a genetically modified mouse model (Johannessen et al., 2008). Like NF1, NF2 also can regulate AKT/mTOR signaling (Scoles, 2008); the NF2-encoded protein, merlin, does so by binding to PIKE (phosphatidylinositol 3-kinase enhancer).
Germline inactivation of the von Hippel-Lindau tumor suppressor gene (VHL) causes hamartomatous tumors associated with the von-Hippel-Lindau syndrome. Moreover, most renal cell carcinomas have biallelic alterations in the von VHL gene, resulting in the accumulation of hypoxia-inducible factors 1 and 2 , and downstream targets including vascular endothelial growth factor (VEGF) (Cho et al., 2007). The observed clinical efficacy of mTOR inhibitors in patients with renal cell carcinoma may be mediated in part by the dependence of efficient hypoxia-inducible factor translation.
Hybrid gene
A schematic model of mTOR signaling depicts various environmental and molecular interactions that influence the pathway. mTOR protein exists in two functionally distinct complexes named mTORC1 and mTORC2. Components are described above, under the Protein Function section. The classical phosphorylation substrates of mTORC1 are S6 kinases and 4E-BP1, although HIF1alpha and PRAS40 also have been shown to have TOR signaling motifs. Because mTOR is shared by both mTORC1 and mTORC2, there may be equilibrium between the two complexes, as well as competition for mTOR (Bhuskar and Hay, 2007). Insulin and other growth factors activate mTORC1 via activation of phosphatidylinositol 3-OH kinase (PI3K) and downstream AKT. Constitutive activation of mTORC1 can occur in the absence of TSC1 or TSC2. Once mTORC1 is activated, it is able to elicit a negative feedback loop to inhibit AKT activity. In opposition, mTORC2 is an activator of AKT, which places this pathway under both positive and negative controls mediated by mTOR. In contrast to growth factor activation of mTORC1, responses to cellular stresses such as energy depletion and amino acid deprivation are mediated by TSC1/2 and/or Rheb (Hay and Sonenberg, 2004; Guertin and Sabatini, 2007). AMPK is activated by reduced intracellular ATP levels and a concomitant increase in intracellular AMP. Under conditions of energy depletion, TSC2 is phosphorylated and activated by AMPK, thereby inhibiting mTORC1 activity. Amino acid starvation also elicits a decrease in mTORC1 activity through Rheb. Abundant evidence suggests that a deregulation between signaling components in the PI3K-AKT-TSC2-Rheb-mTORC1 pathway is a critical step in tumorigenesis. Tumor suppressor genes involved in predisposition to hamartomatous lesions are discussed above in the Implicated Diseases section.
Entity name
Human malignant tumors
Note
Activation of mTOR signaling has been reported in several types of human malignant tumors.
Clinical results have been reported for the mTOR inhibitors CCI-799 (Wyeth), RAD001 (Novartis) and AP23573 (Ariad Pharmaceuticals), all rapamycin analogs (Guertin and Sabatini, 2007). Therapeutic response is highly variable, suggesting that biomarkers still are needed for predicting response to rapamycin therapy. To date, some of the best clinical response rates to rapamycin have been observed in patients suffering from Kaposis sarcoma or mantle-cell lymphoma. Patients with renal cell carcinomas exhibiting a nonclear cell histology also appear to benefit from treatment with mTOR inhibitors (Hanna et al., 2008). Patients with advanced sarcomas are yet another subset of individuals that have benefited from therapeutic mTOR inhibition (Wan and Helman, 2007).
Clinical results have been reported for the mTOR inhibitors CCI-799 (Wyeth), RAD001 (Novartis) and AP23573 (Ariad Pharmaceuticals), all rapamycin analogs (Guertin and Sabatini, 2007). Therapeutic response is highly variable, suggesting that biomarkers still are needed for predicting response to rapamycin therapy. To date, some of the best clinical response rates to rapamycin have been observed in patients suffering from Kaposis sarcoma or mantle-cell lymphoma. Patients with renal cell carcinomas exhibiting a nonclear cell histology also appear to benefit from treatment with mTOR inhibitors (Hanna et al., 2008). Patients with advanced sarcomas are yet another subset of individuals that have benefited from therapeutic mTOR inhibition (Wan and Helman, 2007).
Disease
Collectively, there are a number of mechanisms that contribute to the deregulation of the AKT/mTOR pathway in human malignant tumors (Altomare and Testa, 2005; Wan and Helman, 2007). Phospho-AKT immunohistochemical staining is frequently associated with phospho-mTOR staining. mTOR has emerged as a validated therapeutic target in cancer (Abraham and Eng, 2008).
Specific to mTOR, mTORC2 activity was found to be elevated in glioma cell lines and primary tumors as compared with normal brain tissue (Masri et al., 2007). Overexpression of Rictor increased mTORC2 activity, anchorage-independent growth in soft agar, S-phase cell cycle distribution, motility, and integrin expression, whereas knockdown of Rictor inhibited these events. Xenograft studies also supported a role for increased mTORC2 activity in tumorigenesis and enhanced tumor growth. PKCalpha activity was shown to be dependent of Rictor-expression, consistent with the known regulation of actin organization by mTORC2 via PKCalpha. Collectively, these data suggest that mTORC2 is hyperactivated in gliomas and promotes tumor cell proliferation and invasive potential due to increased complex formation in the presence of overexpressed Rictor.
Specific to mTOR, mTORC2 activity was found to be elevated in glioma cell lines and primary tumors as compared with normal brain tissue (Masri et al., 2007). Overexpression of Rictor increased mTORC2 activity, anchorage-independent growth in soft agar, S-phase cell cycle distribution, motility, and integrin expression, whereas knockdown of Rictor inhibited these events. Xenograft studies also supported a role for increased mTORC2 activity in tumorigenesis and enhanced tumor growth. PKCalpha activity was shown to be dependent of Rictor-expression, consistent with the known regulation of actin organization by mTORC2 via PKCalpha. Collectively, these data suggest that mTORC2 is hyperactivated in gliomas and promotes tumor cell proliferation and invasive potential due to increased complex formation in the presence of overexpressed Rictor.
Prognosis
Recent data suggest that inhibition of mTOR results in clinical benefit in patients with poor prognostic features, and in preclinical models this therapeutic effect involves downregulation of HIF1alpha (Hanna et al., 2008).
Entity name
Huntington disease
Note
mTOR has been implicated in Huntington disease, an inherited neurodegenerative disorder.
Disease
Ravikumar et al. (2004) showed that mTOR is sequestered in polyglutamine aggregates in cell models, transgenic mice, and human brains. Sequestration of mTOR impaired its kinase activity and induced autophagy, a key mechanism for clearance of mutant huntingtin fragments to protect against polyglutamine toxicity. Rapamycin also attenuated huntingtin accumulation and cell death in cell models, and inhibited autophagy. Furthermore, rapamycin protected against neurodegeneration in a fly model, and the rapamycin analog CCI-779 decreased aggregate formation in a mouse model of Huntington disease.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 18208369 | 2008 | Mammalian target of rapamycin as a therapeutic target in oncology. | Abraham RT et al |
| 16288292 | 2005 | Perturbations of the AKT signaling pathway in human cancer. | Altomare DA et al |
| 16288294 | 2005 | Tuberous sclerosis complex: linking growth and energy signaling pathways with human disease. | Astrinidis A et al |
| 17041622 | 2006 | Insulin and amino-acid regulation of mTOR signaling and kinase activity through the Rheb GTPase. | Avruch J et al |
| 17419990 | 2007 | The two TORCs and Akt. | Bhaskar PT et al |
| 17255306 | 2007 | The role of mammalian target of rapamycin inhibitors in the treatment of advanced renal cancer. | Cho D et al |
| 8662507 | 1996 | Structure of the FKBP12-rapamycin complex interacting with the binding domain of human FRAP. | Choi J et al |
| 17541983 | 2008 | Pulmonary lymphangioleiomyomatosis (LAM): progress and current challenges. | Goncharova EA et al |
| 17613433 | 2007 | Defining the role of mTOR in cancer. | Guertin DA et al |
| 17920899 | 2007 | Cowden syndrome. | Gustafson S et al |
| 18279068 | 2008 | mTOR pathway in renal cell carcinoma. | Hanna SC et al |
| 15314020 | 2004 | Upstream and downstream of mTOR. | Hay N et al |
| 15624019 | 2005 | Dysregulation of the TSC-mTOR pathway in human disease. | Inoki K et al |
| 18164202 | 2008 | TORC1 is essential for NF1-associated malignancies. | Johannessen CM et al |
| 18177819 | 2008 | Possible mechanisms of disease development in tuberous sclerosis. | Jozwiak J et al |
| 18094094 | 2008 | Smooth muscle-like cells in pulmonary lymphangioleiomyomatosis. | Krymskaya VP et al |
| 16244323 | 2005 | Tuberous sclerosis: a GAP at the crossroads of multiple signaling pathways. | Kwiatkowski DJ et al |
| 17502379 | 2007 | Hypoxia-inducible factor 1alpha is regulated by the mammalian target of rapamycin (mTOR) via an mTOR signaling motif. | Land SC et al |
| 10089303 | 1999 | Refined structure of the FKBP12-rapamycin-FRB ternary complex at 2.2 A resolution. | Liang J et al |
| 18089801 | 2007 | mTORC2 activity is elevated in gliomas and promotes growth and cell motility via overexpression of rictor. | Masri J et al |
| 17517883 | 2007 | The proline-rich Akt substrate of 40 kDa (PRAS40) is a physiological substrate of mammalian target of rapamycin complex 1. | Oshiro N et al |
| 17928127 | 2008 | Regulation of macroautophagy by mTOR and Beclin 1 complexes. | Pattingre S et al |
| 15146184 | 2004 | Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. | Ravikumar B et al |
| 16603397 | 2006 | Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. | Sarbassov DD et al |
| 17980164 | 2008 | The merlin interacting proteins reveal multiple targets for NF2 therapy. | Scoles DR et al |
| 11967149 | 2002 | Identification of a conserved motif required for mTOR signaling. | Schalm SS et al |
| 18030348 | 2007 | PRAS40 and PRR5-like protein are new mTOR interactors that regulate apoptosis. | Thedieck K et al |
| 10749907 | 2000 | National Cancer Institute Workshop Report: the phakomatoses revisited. | Tucker M et al |
| 17277771 | 2007 | Insulin signalling to mTOR mediated by the Akt/PKB substrate PRAS40. | Vander Haar E et al |
| 17766661 | 2007 | The biology behind mTOR inhibition in sarcoma. | Wan X et al |
| 17510057 | 2007 | PRAS40 regulates mTORC1 kinase activity by functioning as a direct inhibitor of substrate binding. | Wang L et al |
| 18281551 | 2008 | Suppression of Peutz-Jeghers polyposis by targeting mammalian target of rapamycin signaling. | Wei C et al |
| 17179228 | 2007 | Rapamycin derivatives reduce mTORC2 signaling and inhibit AKT activation in AML. | Zeng Z et al |
Other Information
Locus ID:
NCBI: 2475
MIM: 601231
HGNC: 3942
Ensembl: ENSG00000198793
Variants:
dbSNP: 2475
ClinVar: 2475
TCGA: ENSG00000198793
COSMIC: MTOR
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000198793 | ENST00000361445 | P42345 |
| ENSG00000198793 | ENST00000376838 | B1AKP8 |
| ENSG00000198793 | ENST00000455339 | B1AKQ2 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA164746311 | everolimus | Chemical | MultilinkAnnotation | associated | 25893859 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 36385451 | 2024 | Aerobic Glycolysis Induced by mTOR/HIF-1α Promotes Early Brain Injury After Subarachnoid Hemorrhage via Activating M1 Microglia. | 6 |
| 37578594 | 2024 | Loss of RACK1 promotes glutamine addiction via activating AKT/mTOR/ASCT2 axis to facilitate tumor growth in gastric cancer. | 3 |
| 37592783 | 2024 | mirna-383-5p Functions as an Anti-oncogene in Glioma through the Akt/mTOR Signaling Pathway by Targeting VEGFA. | 0 |
| 37731335 | 2024 | AFF4 regulates osteogenic potential of human periodontal ligament stem cells via mTOR-ULK1-autophagy axis. | 1 |
| 37739092 | 2024 | Melatonin inhibits fibroblast cell functions and hypertrophic scar formation by enhancing autophagy through the MT2 receptor-inhibited PI3K/Akt /mTOR signaling. | 1 |
| 37773574 | 2024 | Gene expression of protein kinase AMP-activated catalytic subunit alpha 1 (PRKAA1), solute carrier family 2 member 1 (SLC2A1) and mechanistic target of rapamycin (MTOR) in metformin-treated type 2 diabetes patients with COVID-19: impact on inflammation markers. | 2 |
| 37806601 | 2024 | Assessing the role of phosphorylated S6 ribosomal protein in the pathological diagnosis of pulmonary antibody-mediated rejection. | 1 |
| 37812329 | 2024 | mTOR Plays an Important Role in the Stemness of Human Fetal Cartilage Progenitor Cells (hFCPCs). | 0 |
| 37850256 | 2024 | CCL2 promotes metastasis and epithelial-mesenchymal transition of non-small cell lung cancer via PI3K/Akt/mTOR and autophagy pathways. | 4 |
| 37889103 | 2024 | Canonical and Nuclear mTOR Specify Distinct Transcriptional Programs in Androgen-Dependent Prostate Cancer Cells. | 1 |
| 37909722 | 2024 | KIRREL promotes the proliferation of gastric cancer cells and angiogenesis through the PI3K/AKT/mTOR pathway. | 0 |
| 37955633 | 2024 | TLR/mTOR inflammatory signaling pathway: novel insight for the treatment of schizophrenia. | 2 |
| 37956909 | 2024 | Branched-chain amino acid catabolic defect in vascular smooth muscle cells drives thoracic aortic dissection via mTOR hyperactivation. | 2 |
| 37971211 | 2024 | Nasopharyngeal carcinoma with non-squamous phenotype may be a variant of nasopharyngeal squamous cell carcinoma after inhibition of EGFR/PI3K/AKT/mTOR pathway. | 0 |
| 37982952 | 2024 | Transferrin receptor 1 promotes hepatocellular carcinoma progression and metastasis by activating the mTOR signaling pathway. | 0 |
Citation
Deborah A Altomare ; Joseph R Testa
MTOR (FK506 binding protein 12-rapamycin associated protein 1)
Atlas Genet Cytogenet Oncol Haematol. 2008-06-01
Online version: http://atlasgeneticsoncology.org/gene/40639/mtor
