CFLAR (CASP8 and FADD-like apoptosis regulator)
2008-09-01 Christophe Le Clorennec , Daniel B Longley , Timothy Wilson AffiliationDrug Resistance Group, Centre for Cancer Research, Cell Biology, Queens University Belfast, Belfast, Northern Ireland
Identity
DNA/RNA
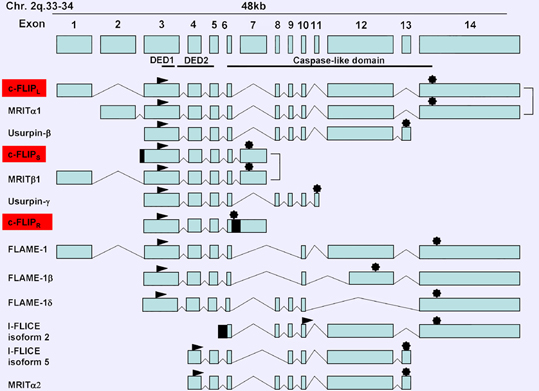
Description
Transcription
Proteins
Note
Soon after the discovery of v-FLIP proteins, the mammalian cellular counterparts were identified, and called c-FLIP proteins (also called CASH, Casper, CLARP, FLAME, I-FLICE, MRIT or usurpin). Among 13 distinct c-FLIP splice variants which have been reported, only three are expressed as proteins: the 55 kDa long form (c-FLIPL), the 26 kDa short form (c-FLIPS) and the 24 kDa short form of c-FLIP (c-FLIPR), identified in the Raji B-cell line (Golks et al., 2005; Budd et al., 2006).

Description
The short form c-FLIPS is composed of 221 amino acids and has the same structure as vFLIP proteins, except that in addition to the two DEDs of cFLIPS, a carboxy-terminal tail composed of approximatively 20 amino acids is present that seems to be crucial for its ubiquitinylation and subsequent proteasomal degradation (Poukkula et al., 2005).
The short form c-FLIPR is composed of 213 amino acids, contains two DEDs and lacks the additional carboxy terminal amino acids present in c-FLIPS (Golks et al., 2005).
Expression
Localisation
Function
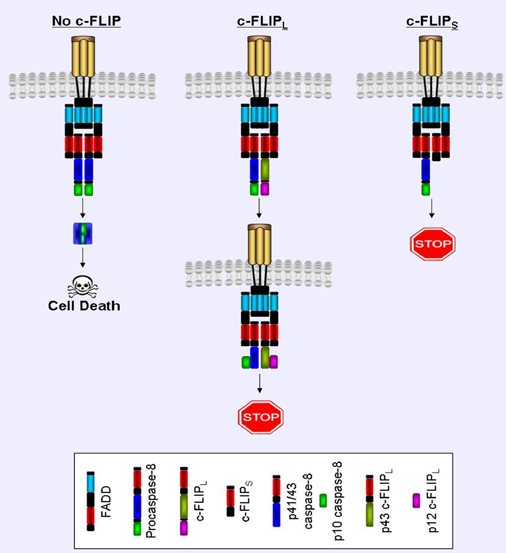
When the death receptors are stimulated by their corresponding ligand, they recruit the adapter molecule FADD. FADD can then recruit DED containing proteins, e.g. caspase-8, and form a DISC. c-FLIP inhibits caspase-8 activation at the DISC. c-FLIPL and c-FLIPS have been shown to block death receptor-mediated apoptosis by forming a proteolytically inactive heterodimer with procaspase-8 (Golks et al., 2005; Krueger et al., 2001). However, cleavage is blocked at different stages. For c-FLIPS and c-FLIPR, both cleavage steps required for procaspase-8 activation are completely blocked. In contrast, c-FLIPL allows partial cleavage of procaspase-8 at the DISC (Figure 3). When a molecule of procaspase-8 and c-FLIPL come into contact at the DISC, a conformational change in the two molecules occurs. This leads to the autocatalytic cleavage of the p10 subunit from procaspase-8. c-FLIPL is also partially cleaved by the procaspase-8 molecule to generate a p12 subunit. However, cleavage is stopped at this stage and no p18 subunit is generated from caspase-8. It has been hypothesised that the second reciprocal trans-catalytic cleavage step cannot occur because of the lack of the cysteine residue at the active site of c-FLIPL (Micheau, 2002). The resulting cleavage products are p41/43- and p10-caspase-8 products; and p43- and p12-c-FLIPL intermediates. Furthermore, Kreuger et al demonstrated that the p41/43-caspase-8 and p43-c-FLIPL intermediates remain bound at the DISC (Krueger et al., 2001). Recently, it has been proposed that the DISC-bound caspase 8/FLIP complex has catalytic activity that is not capable of generating a pro-apoptotic signal, but that can cleave local substrates such as RIP (receptor-interacting protein) (Micheau, 2002).
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 15258564 | 2004 | Possible role of FLICE-like inhibitory protein (FLIP) in chemoresistant ovarian cancer cells in vitro. | Abedini MR et al |
| 10209153 | 1999 | Apoptosis control by death and decoy receptors. | Ashkenazi A et al |
| 9037025 | 1997 | Death effector domain-containing herpesvirus and poxvirus proteins inhibit both Fas- and TNFR1-induced apoptosis. | Bertin J et al |
| 12477972 | 2002 | Differential sensitivity of endothelial cells of various species to apoptosis induced by gene transfer of Fas ligand: role of FLIP levels. | Bouchet D et al |
| 16498450 | 2006 | cFLIP regulation of lymphocyte activation and development. | Budd RC et al |
| 9337844 | 1997 | Caspases: the executioners of apoptosis. | Cohen GM et al |
| 10408805 | 1999 | The immunobiology of multiple sclerosis: an autoimmune disease of the central nervous system. | Conlon P et al |
| 14596792 | 2003 | FLIP protects cardiomyocytes from apoptosis induced by simulated ischemia/reoxygenation, as demonstrated by short hairpin-induced (shRNA) silencing of FLIP mRNA. | Davidson SM et al |
| 12545171 | 2003 | Fas engagement induces neurite growth through ERK activation and p35 upregulation. | Desbarats J et al |
| 11439165 | 2001 | Characterization of the human FLICE-inhibitory protein locus and comparison of the anti-apoptotic activity of four different flip isoforms. | Djerbi M et al |
| 15096587 | 2004 | Expression of the cellular FLICE-inhibitory protein (c-FLIP) protects Hodgkin's lymphoma cells from autonomous Fas-mediated death. | Dutton A et al |
| 11172597 | 2001 | Human pancreatic cancer cells disable function of Fas receptors at several levels in Fas signal transduction pathway. | Elnemr A et al |
| 12700645 | 2003 | FLIP is expressed in mouse testis and protects germ cells from apoptosis. | Giampietri C et al |
| 15701649 | 2005 | c-FLIPR, a new regulator of death receptor-induced apoptosis. | Golks A et al |
| 9743343 | 1998 | Intracellular regulation of TRAIL-induced apoptosis in human melanoma cells. | Griffith TS et al |
| 10442092 | 1999 | The modular nature of apoptotic signaling proteins. | Hofmann K et al |
| 9092488 | 1997 | A novel family of viral death effector domain-containing molecules that inhibit both CD-95- and tumor necrosis factor receptor-1-induced apoptosis. | Hu S et al |
| 9217161 | 1997 | Inhibition of death receptor signals by cellular FLIP. | Irmler M et al |
| 10837247 | 2000 | The caspase-8 inhibitor FLIP promotes activation of NF-kappaB and Erk signaling pathways. | Kataoka T et al |
| 15024054 | 2004 | N-terminal fragment of c-FLIP(L) processed by caspase 8 specifically interacts with TRAF2 and induces activation of the NF-kappaB signaling pathway. | Kataoka T et al |
| 9126933 | 1997 | Differential induction of apoptosis by Fas-Fas ligand interactions in human monocytes and macrophages. | Kiener PA et al |
| 11897874 | 2002 | Human CD34+ hematopoietic stem/progenitor cells express high levels of FLIP and are resistant to Fas-mediated apoptosis. | Kim H et al |
| 10690508 | 2000 | Molecular determinants of response to TRAIL in killing of normal and cancer cells. | Kim K et al |
| 15183989 | 2004 | c-FLIP expression in bladder urothelial carcinomas: its role in resistance to Fas-mediated apoptosis and clinicopathologic correlations. | Korkolopoulou P et al |
| 17589543 | 2007 | Life and death in peripheral T cells. | Krammer PH et al |
| 11279218 | 2001 | Cellular FLICE-inhibitory protein splice variants inhibit different steps of caspase-8 activation at the CD95 death-inducing signaling complex. | Krueger A et al |
| 12078890 | 2002 | Biology of Hodgkin's lymphoma. | Küppers R et al |
| 16247474 | 2006 | c-FLIP inhibits chemotherapy-induced colorectal cancer cell death. | Longley DB et al |
| 12360407 | 2002 | Mechanisms of resistance to TRAIL-induced apoptosis in primary B cell chronic lymphocytic leukaemia. | MacFarlane M et al |
| 12060768 | 2002 | FLIP switches Fas-mediated glucose signaling in human pancreatic beta cells from apoptosis to cell replication. | Maedler K et al |
| 15507484 | 2004 | FLICE/caspase-8 activation triggers anoikis induced by beta1-integrin blockade in human keratinocytes. | Marconi A et al |
| 15078899 | 2004 | c-FLIP mediates resistance of Hodgkin/Reed-Sternberg cells to death receptor-induced apoptosis. | Mathas S et al |
| 15297424 | 2004 | CD95-mediated apoptosis is impaired at receptor level by cellular FLICE-inhibitory protein (long form) in wild-type p53 human ovarian carcinoma. | Mezzanzanica D et al |
| 12215447 | 2002 | The long form of FLIP is an activator of caspase-8 at the Fas death-inducing signaling complex. | Micheau O et al |
| 14662022 | 2003 | Upregulation of FLIP(S) by Akt, a possible inhibition mechanism of TRAIL-induced apoptosis in human gastric cancers. | Nam SY et al |
| 11753607 | 2001 | Sensitization to TRAIL-induced apoptosis and modulation of FLICE-inhibitory protein in B chronic lymphocytic leukemia by actinomycin D. | Olsson A et al |
| 11741321 | 2001 | Alternative splicing variants of c-FLIP transduce the differential signal through the Raf or TRAF2 in TNF-induced cell proliferation. | Park SJ et al |
| 15886205 | 2005 | Rapid turnover of c-FLIPshort is determined by its unique C-terminal tail. | Poukkula M et al |
| 11104808 | 2000 | Fas engagement induces the maturation of dendritic cells (DCs), the release of interleukin (IL)-1beta, and the production of interferon gamma in the absence of IL-12 during DC-T cell cognate interaction: a new role for Fas ligand in inflammatory responses. | Rescigno M et al |
| 15367674 | 2004 | Direct repression of FLIP expression by c-myc is a major determinant of TRAIL sensitivity. | Ricci MS et al |
| 17513603 | 2007 | Cellular FLICE-inhibitory protein regulates chemotherapy-induced apoptosis in breast cancer cells. | Rogers KM et al |
| 11329136 | 2001 | Increased expression of cFLIP(L) in colonic adenocarcinoma. | Ryu BK et al |
| 9880531 | 1999 | The role of c-FLIP in modulation of CD95-induced apoptosis. | Scaffidi C et al |
| 12393731 | 2003 | Loss of the B-lineage-specific gene expression program in Hodgkin and Reed-Sternberg cells of Hodgkin lymphoma. | Schwering I et al |
| 10074154 | 1999 | Sequence and genomic analysis of a Rhesus macaque rhadinovirus with similarity to Kaposi's sarcoma-associated herpesvirus/human herpesvirus 8. | Searles RP et al |
| 11164911 | 2001 | Overexpression of the apoptosis inhibitor FLIP in T cells correlates with disease activity in multiple sclerosis. | Semra YK et al |
| 11063839 | 2000 | Increased cellular expression of the caspase inhibitor FLIP in intrathecal lymphocytes from patients with multiple sclerosis. | Sharief MK et al |
| 9228018 | 1997 | FLAME-1, a novel FADD-like anti-apoptotic molecule that regulates Fas/TNFR1-induced apoptosis. | Srinivasula SM et al |
| 11101869 | 2000 | Control of target cell survival in thyroid autoimmunity by T helper cytokines via regulation of apoptotic proteins. | Stassi G et al |
| 11943736 | 2002 | Constitutive expression of c-FLIP in Hodgkin and Reed-Sternberg cells. | Thomas RK et al |
| 9087414 | 1997 | Viral FLICE-inhibitory proteins (FLIPs) prevent apoptosis induced by death receptors. | Thome M et al |
| 17785559 | 2007 | Overexpression of FLIPL is an independent marker of poor prognosis in colorectal cancer patients. | Ullenhag GJ et al |
| 17433827 | 2007 | The relationship between c-FLIP expression and human papillomavirus E2 gene disruption in cervical carcinogenesis. | Wang W et al |
| 17575142 | 2007 | c-FLIP: a key regulator of colorectal cancer cell death. | Wilson TR et al |
| 15466204 | 2004 | Persistent c-FLIP(L) expression is necessary and sufficient to maintain resistance to tumor necrosis factor-related apoptosis-inducing ligand-mediated apoptosis in prostate cancer. | Zhang X et al |
| 14636156 | 2004 | Overexpression of cellular FLICE-inhibitory protein (FLIP) in gastric adenocarcinoma. | Zhou XD et al |
Other Information
Locus ID:
NCBI: 8837
MIM: 603599
HGNC: 1876
Ensembl: ENSG00000003402
Variants:
dbSNP: 8837
ClinVar: 8837
TCGA: ENSG00000003402
COSMIC: CFLAR
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38710704 | 2024 | Deciphering DED assembly mechanisms in FADD-procaspase-8-cFLIP complexes regulating apoptosis. | 0 |
| 38710704 | 2024 | Deciphering DED assembly mechanisms in FADD-procaspase-8-cFLIP complexes regulating apoptosis. | 0 |
| 36400248 | 2023 | The interaction of CFLAR with p130Cas promotes cell migration. | 3 |
| 37591930 | 2023 | cFLIP(S) regulates alternative NLRP3 inflammasome activation in human monocytes. | 2 |
| 36400248 | 2023 | The interaction of CFLAR with p130Cas promotes cell migration. | 3 |
| 37591930 | 2023 | cFLIP(S) regulates alternative NLRP3 inflammasome activation in human monocytes. | 2 |
| 35044632 | 2022 | The deubiquitinase Usp27x as a novel regulator of cFLIP(L) protein expression and sensitizer to death-receptor-induced apoptosis. | 5 |
| 35115486 | 2022 | cFLIP downregulation is an early event required for endoplasmic reticulum stress-induced apoptosis in tumor cells. | 9 |
| 35422085 | 2022 | c-FLIP promotes drug resistance in non-small-cell lung cancer cells via upregulating FoxM1 expression. | 3 |
| 35044632 | 2022 | The deubiquitinase Usp27x as a novel regulator of cFLIP(L) protein expression and sensitizer to death-receptor-induced apoptosis. | 5 |
| 35115486 | 2022 | cFLIP downregulation is an early event required for endoplasmic reticulum stress-induced apoptosis in tumor cells. | 9 |
| 35422085 | 2022 | c-FLIP promotes drug resistance in non-small-cell lung cancer cells via upregulating FoxM1 expression. | 3 |
| 33837174 | 2021 | Cellular FLICE-like inhibitory protein (cFLIP) critically maintains apoptotic resistance in human lens epithelial cells. | 4 |
| 34302634 | 2021 | Lactucin induces apoptosis through reactive oxygen species-mediated BCL-2 and CFLAR(L) downregulation in Caki-1 cells. | 2 |
| 33837174 | 2021 | Cellular FLICE-like inhibitory protein (cFLIP) critically maintains apoptotic resistance in human lens epithelial cells. | 4 |
Citation
Christophe Le Clorennec ; Daniel B Longley ; Timothy Wilson
CFLAR (CASP8 and FADD-like apoptosis regulator)
Atlas Genet Cytogenet Oncol Haematol. 2008-09-01
Online version: http://atlasgeneticsoncology.org/gene/40065/cflar
