RASSF2 (Ras association (RalGDS/AF-6) domain family member 2)
2009-08-01 Luke B Hesson , Farida Latif AffiliationIdentity
DNA/RNA
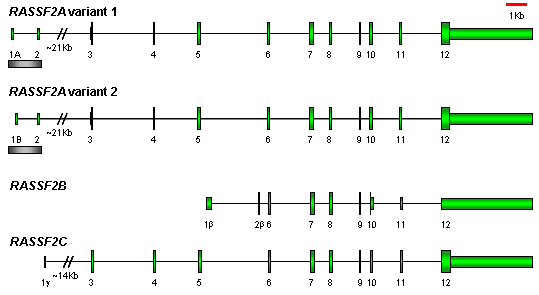
Description
Proteins
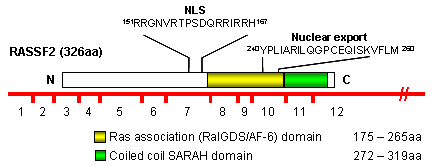
Description
Expression
Localisation
Function
Similar to several other RASSF members RASSF2 suppresses tumour growth when expressed. This has been demonstrated for colorectal, lung, breast, gastric, nasopharyngeal and oral squamous cell carcinoma (OSCC) cell lines in vitro using colony formation, growth curve and soft agar growth assays (Akino et al., 2005; Vos et al., 2003; Cooper et al., 2008; Maruyama et al., 2008; Imai et al., 2008; Zhang et al., 2006). Furthermore, RASSF2 re-expression in breast tumour cells inhibits in vivo tumour growth when cells are subcutaneously injected into severe combined immunodeficiency (SCID) mice (Cooper et al., 2008). Several studies demonstrate that these tumour suppressive properties are likely to arise from the ability of RASSF2 to regulate apoptosis and cell cycle progression (Vos et al., 2003; Maruyama et al., 2008; Imai et al., 2008; Akino et al., 2005). In breast cancer cells and Cos-7 cells growth suppression by RASSF2 is dependent on the nuclear localisation signal (NLS) located at amino acids 151-167 (Cooper et al., 2008; Kumari et al., 2009), whilst other reports have indicated that in OSCC and gastric cancer cells the C-terminal portion of RASSF2 (RASSF2 [163-326]), containing the RA domain, is critical for tumour suppressive function (Imai et al., 2008; Maruyama et al., 2008). Interestingly, in OSCC this C-terminal portion exhibited enhanced growth suppression relative to full length RASSF2 (Imai et al., 2008). In fact, RASSF2 [163-326] also disrupts the NLS yet leaves the sequence required for nuclear export intact. In a separate study of colorectal cancer cells both RASSF2 truncations (RASSF2 [1-163] and RASSF2 [163-326]) exhibited reduced growth suppression compared to full length RASSF2 (Akino et al., 2005). Whilst in gastric cancer transfection of RASSF2 with deletion of the NLS [RASSF2deltaNLS] actually increased the percentage of apoptotic cells relative to full length RASSF2 (Maruyama et al., 2008). These studies indicate the growth suppressive properties of RASSF2 are likely cell background specific but more importantly that nuclear import, nuclear export and the Ras-association domain are required for correctly regulated RASSF2 growth suppression.
RASSF2 interaction with Ras
RASSF2 contains a functional RA domain that displays a strong binding to K-Ras, but only weak binding to H-Ras (Vos et al., 2003). RASSF2 associates with the K-Ras effector domain in a GTP-dependent manner thus displaying the basic properties of a genuine Ras effector. RASSF2 growth inhibition is enhanced in the presence of K-RasG12V. Furthermore, siRNA-mediated knock-down of RASSF2 in K-Ras transformed cells enhanced anchorage-independent growth. However, in the absence of K-Ras transformation knock-down of RASSF2 inhibited growth (Akino et al., 2005). These data indicate that RASSF2 mediates some of the growth inhibitory properties of K-Ras and that inactivation of RASSF2 enhances K-Ras-induced transformation.
RASSF2 interaction with the proapoptotic kinases MST1 and MST2
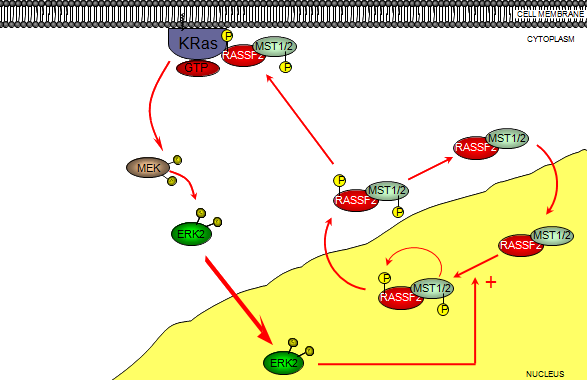
How the interaction of RASSF proteins with Ras results in growth suppression has been the subject of intense investigation. The proapoptotic mammalian Serine/Threonine kinases MST1 and MST2 were identified as RASSF2 interacting partners by yeast two-hybrid (Y2-H) (Khokhlatchev et al., 2002). RASSF1 and RASSF5 were also identified as MST binding partners as part of a novel Ras-regulated signalling pathway. Recently the interaction of RASSF2 and MST1/2 was formally demonstrated in human cells at the endogenous level. Interaction occurs between the SARAH domains found within RASSF2 and MST1/2 (Cooper et al., 2009). RASSF2 appears to have a distinct role in regulating MST2 function. Activation of MST2 is followed by a rapid proteasome-dependent loss of MST2 stability (that is not associated with MST2 cleavage). Interestingly, over expression of RASSF2 results in increased levels of MST2 and provides protection of MST2 from degradation following its activation. In agreement with this loss of RASSF2 protein in colorectal tumours, or in colorectal tumour cell lines in which RASSF2 levels are decreased by shRNAi, also leads to decreases in MST2 levels (Cooper et al., 2009). RASSF2 appears to be a substrate for MST1 and MST2 and co-expression of either kinase with RASSF2 relocalises RASSF2 from the nucleus to the cytoplasm in a manner dependent on kinase activity (Cooper et al., 2009). Since MST2 remains in complex with RASSF2 following its activation these data collectively suggest RASSF2 stabilises active MST2 allowing (or perhaps even targeting) MST2 substrate phosphorylation. This RASSF2-mediated stabilisation also appears to be true for MST1. Thus loss of RASSF2, as is frequently observed in cancer, leads to loss of MST1 and MST2 leading to a decrease in apoptotic potential. That RASSF2 appears to be capable of influencing MST stability so drastically is likely to be due to the observation that the majority of both MST1 and MST2 are in complex with RASSF2 in at least some cell types (Cooper et al., 2009). The interaction of RASSF2 with MST1/2 poses an interesting question with regards to the regulation of RASSF2 localisation. Both RASSF2 and MST1/2 have been shown to contain sequences essential for nuclear import and export (Lee and Yonehara, 2002; Kumari et al., 2009). Both the NLS and NES sequences within MST1/2 and RASSF2 respectively are located very close to or within the SARAH domains and neither are canonical NLS/NES sequences. Mapping of both these sequences were determined by deletion mapping, which would most likely also affect RASSF2-MST1/2 interaction thus it now seems likely that the RASSF2-MST1/2 complex constantly cycles between the nucleus (by virtue of RASSF2 NLS) and cytoplasm (by virture of MST1/2 NES) and disruption of the interaction between RASSF2 and MST1/2 would likely affect the localisation of both proteins. Also, the fact that RASSF2 translocation to the cytoplasm is dependent on ERK2 activity (Kumari and Mahalingam, 2009) suggests the Ras-MEK-ERK pathway may serve to phosphorylate MST1/2, which then phosphorylates RASSF2, translocating it to the cytoplasm and allowing RASSF2 to interact with Ras (figure 4). That nuclear RASSF2 is required for full tumour suppressor activity (Cooper et al., 2008; Kumari et al., 2009) may be explained by the fact that ERK2 translocates to the nucleus upon its activation (Khokhlatchev et al., 1998).
Other functions of RASSF2
Other functions and interacting partners of RASSF2 are extremely likely. Y2-H using RASSF2 as bait implicates NORE1A and RASSF3 in RASSF2 function, although these have not yet been confirmed in mammalian cells (Hesson et al., 2005). These interactions may implicate other RASSF members in modulating RASSF2 function and suggests a complex network of cross-talk between signalling pathways involving RASSF proteins. Also, the exact mechanisms of apoptotic and cell cycle regulation of RASSF2 have yet to be completely defined. Microarray analysis of gene expression before and after exogenous expression of RASSF2 in gastric and OSCC cancer cell lines showed RASSF2 downregulates expression of several inflammatory response genes including the cytokines IL-8, LCN2, CXCL1, CXCL2, CXCL3, CXCL5 and CXCL6, CCL20 and CCL21 and genes involved in immune-cell chemotaxis (Maruyama et al., 2008; Imai et al., 2008). A possible pathway influenced by RASSF2 is the NF-kB pathway since over expression of RASSF2 significantly downregulated NF-kB transcriptional activity (Maruyama et al., 2008; Imai et al., 2008). Of note is the recent observation that pigs experimentally infected with Porcine Circovirus Type 2 (PCV2) show upregulation of several CXCL family cytokines as well as RASSF2 (Fernandes et al., 2009) therefore it is likely a role for RASSF2 in regulating immune response pathways remains to be discovered. There is also evidence that RASSF2 may regulate the actin cytoskeleton since re-expression of RASSF2 leads to loss of stress fibres, cell rounding and the suppression of RhoGTPase activation (Maruyama et al., 2008; Akino et al., 2005). Additionally, RASSF2 upregulation appears to be a cellular response to ionising radiation (Sakamoto-Hojo et al., 2003).
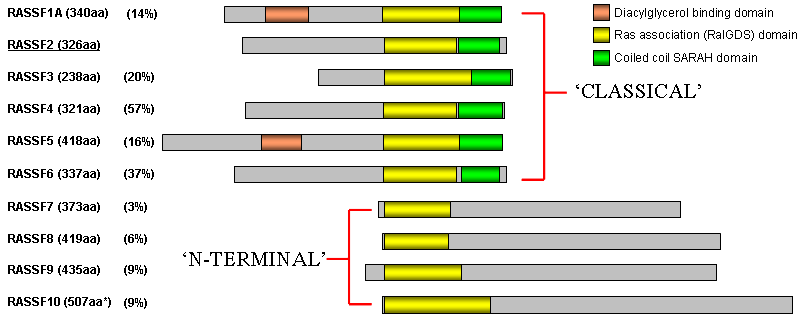
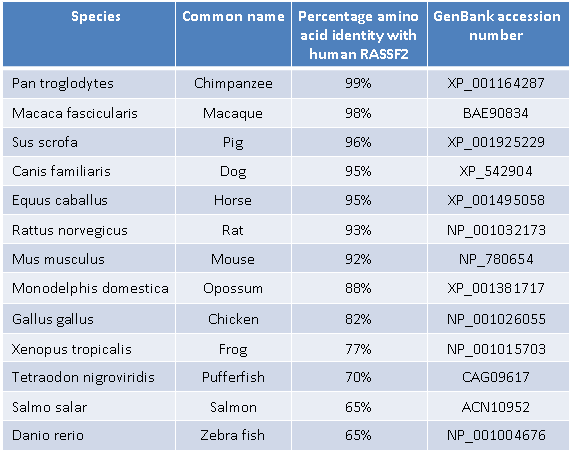
Homology
Mutations
Note
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16012945 | 2005 | The Ras effector RASSF2 is a novel tumor-suppressor gene in human colorectal cancer. | Akino K et al |
| 17404571 | 2007 | RASSF6 is a novel member of the RASSF family of tumor suppressors. | Allen NP et al |
| 16460908 | 2006 | Comparative genomic organization of the human and bovine PRNP locus. | Choi SH et al |
| 15375500 | 2004 | Aberrant methylation of RASSF4/AD037 in nasopharyngeal carcinoma. | Chow LS et al |
| 17891178 | 2008 | Epigenetic regulation of the ras effector/tumour suppressor RASSF2 in breast and lung cancer. | Cooper WN et al |
| 19525978 | 2009 | RASSF2 associates with and stabilizes the proapoptotic kinase MST2. | Cooper WN et al |
| 15574778 | 2004 | RASSF4/AD037 is a potential ras effector/tumor suppressor of the RASSF family. | Eckfeld K et al |
| 16265349 | 2005 | RASSF2, a potential tumour suppressor, is silenced by CpG island hypermethylation in gastric cancer. | Endoh M et al |
| 19544206 | 2009 | Exploratory study on the transcriptional profile of pigs subclinically infected with porcine circovirus type 2. | Fernandes LT et al |
| 17923875 | 2007 | Genetic and epigenetic alterations of Ras signalling pathway in colorectal neoplasia: analysis based on tumour clinicopathological features. | Harada K et al |
| 17325427 | 2007 | The role of RASSF1A methylation in cancer. | Hesson LB et al |
| 19570220 | 2009 | The novel RASSF6 and RASSF10 candidate tumour suppressor genes are frequently epigenetically inactivated in childhood leukaemias. | Hesson LB et al |
| 15806169 | 2005 | CpG island promoter hypermethylation of a novel Ras-effector gene RASSF2A is an early event in colon carcinogenesis and correlates inversely with K-ras mutations. | Hesson LB et al |
| 19509163 | 2009 | Methylation of RASSF1A, RASSF2A, and HIN-1 is associated with poor outcome after radiotherapy, but not surgery, in oral squamous cell carcinoma. | Huang KH et al |
| 17367779 | 2007 | Ras-association domain family protein 6 induces apoptosis via both caspase-dependent and caspase-independent pathways. | Ikeda M et al |
| 18294275 | 2008 | Epigenetic inactivation of RASSF2 in oral squamous cell carcinoma. | Imai T et al |
| 17549418 | 2007 | Epigenetic inactivation of the RAS-effector gene RASSF2 in lung cancers. | Kaira K et al |
| 17950780 | 2008 | CpG island methylation is frequently present in tubulovillous and villous adenomas and correlates with size, site, and villous component. | Kakar S et al |
| 19478941 | 2009 | High mutability of the tumor suppressor genes RASSF1 and RBSP3 (CTDSPL) in cancer. | Kashuba VI et al |
| 11864565 | 2002 | Identification of a novel Ras-regulated proapoptotic pathway. | Khokhlatchev A et al |
| 19555684 | 2009 | Extracellular signal-regulated kinase 2 (ERK-2) mediated phosphorylation regulates nucleo-cytoplasmic shuttling and cell growth control of Ras-associated tumor suppressor protein, RASSF2. | Kumari G et al |
| 17320110 | 2007 | Nuclear transport of Ras-associated tumor suppressor proteins: different transport receptor binding specificities for arginine-rich nuclear targeting signals. | Kumari G et al |
| 15586366 | 2005 | Analysis of ovarian cancer cell lines using array-based comparative genomic hybridization. | Lambros MB et al |
| 11805089 | 2002 | Phosphorylation and dimerization regulate nucleocytoplasmic shuttling of mammalian STE20-like kinase (MST). | Lee KK et al |
| 19155294 | 2009 | Consistent deregulation of gene expression between human and murine MLL rearrangement leukemias. | Li Z et al |
| 18404674 | 2008 | Hypermethylation of RAS effector related genes and DNA methyltransferase 1 expression in endometrial carcinogenesis. | Liao X et al |
| 18310659 | 2008 | Cytoplasmic RASSF2A is a proapoptotic mediator whose expression is epigenetically silenced in gastric cancer. | Maruyama R et al |
| 16469793 | 2006 | Extensive DNA methylation in normal colorectal mucosa in hyperplastic polyposis. | Minoo P et al |
| 18161048 | 2008 | Aberrant methylation of multiple tumor suppressor genes in aging liver, chronic hepatitis, and hepatocellular carcinoma. | Nishida N et al |
| 17183069 | 2007 | Genetic and epigenetic profiling in early colorectal tumors and prediction of invasive potential in pT1 (early invasive) colorectal cancers. | Nosho K et al |
| 16096369 | 2005 | High frequency somatic mutations in RASSF1A in nasopharyngeal carcinoma. | Pan ZG et al |
| 19459176 | 2009 | DNA methylation biomarkers of prostate cancer: confirmation of candidates and evidence urine is the most sensitive body fluid for non-invasive detection. | Payne SR et al |
| 14644343 | 2003 | Gene expression profiles in human cells submitted to genotoxic stress. | Sakamoto-Hojo ET et al |
| 16344548 | 2006 | The RASSF1A tumor suppressor activates Bax via MOAP-1. | Vos MD et al |
| 12732644 | 2003 | RASSF2 is a novel K-Ras-specific effector and potential tumor suppressor. | Vos MD et al |
| 17013896 | 2007 | Inactivation of RASSF2A by promoter methylation correlates with lymph node metastasis in nasopharyngeal carcinoma. | Zhang Z et al |
Other Information
Locus ID:
NCBI: 9770
MIM: 609492
HGNC: 9883
Ensembl: ENSG00000101265
Variants:
dbSNP: 9770
ClinVar: 9770
TCGA: ENSG00000101265
COSMIC: RASSF2
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000101265 | ENST00000379376 | P50749 |
| ENSG00000101265 | ENST00000379400 | P50749 |
Expression (GTEx)
Pathways
| Pathway | Source | External ID |
|---|---|---|
| Hippo signaling pathway -multiple species | KEGG | ko04392 |
| Hippo signaling pathway -multiple species | KEGG | hsa04392 |
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38036076 | 2024 | Hyperhomocysteinemia may aggravate abdominal aortic aneurysm formation by up-regulating RASSF2. | 0 |
| 38493315 | 2024 | Diagnostic Value of GSTP1, RASSF1, AND RASSF2 Methylation in Serum of Prostate Cancer Patients. | 0 |
| 38036076 | 2024 | Hyperhomocysteinemia may aggravate abdominal aortic aneurysm formation by up-regulating RASSF2. | 0 |
| 38493315 | 2024 | Diagnostic Value of GSTP1, RASSF1, AND RASSF2 Methylation in Serum of Prostate Cancer Patients. | 0 |
| 32029705 | 2020 | The RUNX1-ETO target gene RASSF2 suppresses t(8;21) AML development and regulates Rac GTPase signaling. | 8 |
| 33051258 | 2020 | RASSF effectors couple diverse RAS subfamily GTPases to the Hippo pathway. | 14 |
| 32029705 | 2020 | The RUNX1-ETO target gene RASSF2 suppresses t(8;21) AML development and regulates Rac GTPase signaling. | 8 |
| 33051258 | 2020 | RASSF effectors couple diverse RAS subfamily GTPases to the Hippo pathway. | 14 |
| 30992382 | 2019 | CircRASSF2 promotes laryngeal squamous cell carcinoma progression by regulating the miR-302b-3p/IGF-1R axis. | 48 |
| 31453724 | 2019 | Association Between RASSF2 Methylation and Gastric Cancer: A PRISMA-Compliant Systematic Review and Meta-Analysis. | 1 |
| 30992382 | 2019 | CircRASSF2 promotes laryngeal squamous cell carcinoma progression by regulating the miR-302b-3p/IGF-1R axis. | 48 |
| 31453724 | 2019 | Association Between RASSF2 Methylation and Gastric Cancer: A PRISMA-Compliant Systematic Review and Meta-Analysis. | 1 |
| 29504361 | 2018 | Relationship between methylation status of RASSF2A gene promoter and endometriosis-associated ovarian cancer. | 2 |
| 29504361 | 2018 | Relationship between methylation status of RASSF2A gene promoter and endometriosis-associated ovarian cancer. | 2 |
| 27901488 | 2017 | Cancer-associated fibroblasts promote cancer cell growth through a miR-7-RASSF2-PAR-4 axis in the tumor microenvironment. | 30 |
Citation
Luke B Hesson ; Farida Latif
RASSF2 (Ras association (RalGDS/AF-6) domain family member 2)
Atlas Genet Cytogenet Oncol Haematol. 2009-08-01
Online version: http://atlasgeneticsoncology.org/gene/43461/rassf2
