THY1 (Thy-1 cell surface antigen)
2010-01-01 John E Bradley , James S Hagood AffiliationDepartments of Pediatrics, Biochemistry, Molecular Genetics, University of Alabama-Birmingham, AL, USA (JEB); Department of Pediatrics, University of California at San Diego, USA (JSH)
DNA/RNA
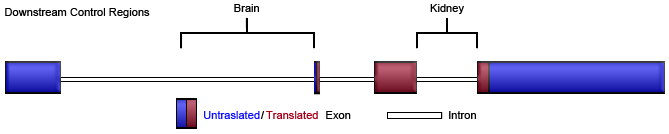
Organization of the human THY1 gene and control elements. There are sequences conferring tissue specificity for the brain in the first intron and kidney in the third intron.
Description
4 exons; DNA size: 5591.
Transcription
Transcription of THY1 is initiated at multiple sites producing an approximately 5591 bp transcript. After splicing, the transcript is reduced to 2143 bp. Transcriptional regulation for tissue-specific expression of Thy-1, as well as for controlling expression in cell sub-populations, and in some cancers, is governed by a number of mechanisms. For tissue-specific transcriptional regulation, the regulatory elements are found exclusively downstream of the transcription initiation site within the gene itself. Differential tissue expression of Thy1 exists between species as closely related as mouse and rat. For example, Thy1 is expressed in mouse thymocytes and splenocytes, but only thymocytes in rat. The absence of Thy1 in rat splenocytes is attributed to a three nucleotide difference in a conserved 36 bp region within the third intron. In mice, this 36 bp region can bind an Ets-l-like nuclear factor expressed by both mouse thymocytes and splenocytes. In rats, the three nucleotide difference renders the 36 bp region no longer capable of binding the similar Ets-l-like nuclear factor expressed by rats. However, rat thymocytes but not splenocytes express another nuclear factor which does recognize the 36 bp region and this is thought to account for the expression of Thy1 in rat thymocytes but not splenocytes. In humans, THY1 is detected in the brain, spleen, kidneys, but not thymus. In mouse, Thy1 is detected in the brain, spleen, thymus, but not kidneys. In transgenic mice, deletion of half the 3 end of intron 1 prevents expression of Thy1 in the brain but allows for its expression in the thymus. The control elements within the first intron of Thy-1 are conserved in human and mouse, as replacement of the first intron in mouse with that from human causes no detectable change in Thy1 expression in the brain. Differences between intron 3 of mouse and human Thy-1 seem to account for expression of Thy-1 in human but not mouse kidney and mouse but not human thymus. In transgenic mice, Thy1 is no longer expressed in the thymus but ectopically expressed in the kidney when the third intron of mouse Thy1 is replaced with the third intron of human THY1. The aforementioned downstream control elements require at a minimum 300 bp of the endogenous Thy1 promoter for transcription to occur. Interestingly, the endogenous Thy1 promoter in itself does not elicit transcription or tissue specificity absent the downstream elements. Conversely, the downstream elements are able to direct tissue-specific transcription of Thy1 with a heterologous promoter. Therefore, the downstream control elements are "promiscuous" with regard to a promoter, while the endogenous promoter is "monogamous" with the downstream control elements. The validity of these assertions is exemplified in the design of the murine thy1.2 genomic expression cassette for driving expression in the nervous system. In this cassette, the coding sequences, as well as the third intron of thy1.2 have all been removed, but the first intron is retained.
Suppression of Thy-1 transcription within sub-populations of lung fibroblasts and the tumorigenic nasopharyngeal cell carcinoma (NPC) cell line HONE1 is shown to occur via hypermethylation of CpG (cytosine-guanine) islands in the Thy-1 gene promoter. Moreover, THY1 is thought to function as a tumor suppressor in NPC as microcell-mediated transfer of an additional intact human chromosome 11 into HONE1 cells decreases colony formation with re-expression of THY1. Tumor segregants of the HONE1 microcell hybrids were all negative for THY1. Concurrently, the region 11q22-23 is shown to be critical for tumorigenicty in NPC. In both rat and human lung fibroblasts, CpG islands in the Thy-1 gene promoter are hypermethylated in the Thy-1 negative fibroblast subpopulation but not in the positive. Thy1 expression is induced in Thy1 (-) fibroblasts by treatment with 5-aza-2-deoxycytidine, a DNA methyltransferase inhibitor. Suppression of THY1 transcription via hypermethylation of its promoter in THY1 (-) lung fibroblasts has implications in the disease idiopathic pulmonary fibrosis (IPF). Fibroblastic foci are populated predominantly by THY1 (-) myofibroblasts and methylation-specific PCR-in situ hybridization has demonstrated THY1 promoters within these areas to be hypermethylated.
Suppression of Thy-1 transcription within sub-populations of lung fibroblasts and the tumorigenic nasopharyngeal cell carcinoma (NPC) cell line HONE1 is shown to occur via hypermethylation of CpG (cytosine-guanine) islands in the Thy-1 gene promoter. Moreover, THY1 is thought to function as a tumor suppressor in NPC as microcell-mediated transfer of an additional intact human chromosome 11 into HONE1 cells decreases colony formation with re-expression of THY1. Tumor segregants of the HONE1 microcell hybrids were all negative for THY1. Concurrently, the region 11q22-23 is shown to be critical for tumorigenicty in NPC. In both rat and human lung fibroblasts, CpG islands in the Thy-1 gene promoter are hypermethylated in the Thy-1 negative fibroblast subpopulation but not in the positive. Thy1 expression is induced in Thy1 (-) fibroblasts by treatment with 5-aza-2-deoxycytidine, a DNA methyltransferase inhibitor. Suppression of THY1 transcription via hypermethylation of its promoter in THY1 (-) lung fibroblasts has implications in the disease idiopathic pulmonary fibrosis (IPF). Fibroblastic foci are populated predominantly by THY1 (-) myofibroblasts and methylation-specific PCR-in situ hybridization has demonstrated THY1 promoters within these areas to be hypermethylated.
Proteins
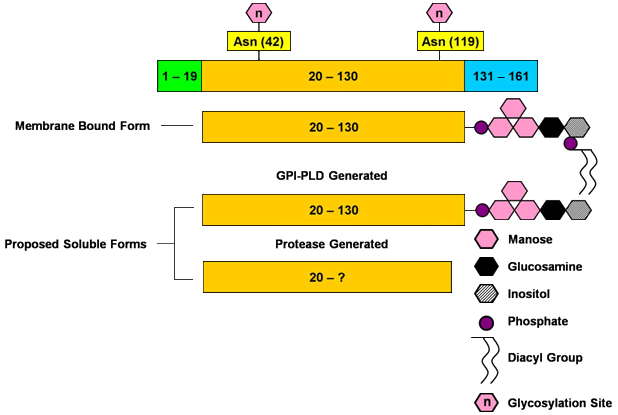
THY1 molecule and proposed soluble forms. THY1 is initially generated as a 161 aa pro form. The initial 19 aa signal peptide is removed, and the terminal 31 aa are replaced with a GPI anchor, generating the mature form, which is anchored to the outer leaflet of the cell membrane by the diacyl group of the GPI anchor. N-linked glycosylation sites depict conserved asparagines within murine Thy1 that are known to be glycosylated. Soluble Thy-1 could be generated either by cleavage of the GPI anchor by GPI-PLD, or by undefined proteases acting at as yet undetermined cleavage sites.
Description
Human and mouse Thy-1 are both initially translated as a 161 and 162 amino acid pro-form, respectively. In mouse, there are two alleles that encode two proteins distinguished by having either arginine or glutamine at position 89. Humans have only one allele of THY1. The first 19 aa of the pro-form are a localization signal that targets it into the ER. Initially, the c-terminal residues 131-161 function as a trans-membrane domain within the ER. Thy-1 undergoes several post translation modifications, including proteolytic cleavage, N-linked glycosylation, and addition of a GPI moiety. The localization signal and trans-membrane domain are proteolytically cleaved away leaving a core protein composed of residues 20-131. The N-linked glycosylation sites of murine Thy1 are at asparagine residues 42, 94, and 118. Only two of these residues are conserved in human at positions 42 and 119. The carbohydrate content of THY1 accounts for nearly 30% of its molecular mass, which ranges from 25 to 37 kDa. Between species, tissue types, and cells in different stages of development: these carbohydrate moieties may vary dramatically. After removal of the trans-membrane domain, a GPI moiety is attached to the c-terminal residue of the core protein, cysteine 130. The GPI moiety contains two fatty-acyl groups that embed into the membrane thereby anchoring Thy-1 to the cell surface. Thus, Thy-1 has no intra-cellular domain. Thy-1 is a member of the immunoglobulin superfamily and as such possesses cysteine residues which form disulfide bonds. A soluble variety of Thy-1 exists and is presumably produced by a proteolytic and/or lipolytic cleavage at the cell surface. If the latter is the case, it is presumed to occur in close proximity to the c-terminus. Completely deglycosylated membranous and soluble THY1 have indistinguishable migration speeds through a polyacrylamide gel.
Expression
In mouse and human, Thy-1 is expressed at the cell surface of mature neurons, a subset of fibroblasts, and activated natural killer cells. With the exception of these cell types, mouse and human each have unique Thy-1 expression profiles. Thy1 covers up to 10-20% of mouse thymocyte cell surface but levels diminish in locations of greater thymocyte maturation. Specifically, cortical thymocytes express higher levels of Thy1 than medullary thymocytes and Lymph node cells have levels less than both. The only human thymocytes to express THY1 are a small population of cortical thymocytes, whereas expression of Thy1 in the mouse is broader and also includes peripheral T cells. In humans, THY1 is also expressed by endothelial cells (conditionally), smooth muscle cells, some glial cells, a subset of CD34 (+) bone marrow cells, and umbilical cord blood- and fetal liver-derived hematopoietic stem cells.
A soluble variety of THY1 is detectable in serum, cerebral spinal fluid, wound fluid from venous leg ulcers, and the synovial fluid from joints in rheumatoid arthritis. Cultured lung fibroblasts shed THY1 into the media when treated with pro-inflammatory cyokines, such as IL-1beta and TNF-alpha. The opposite effect is elicited in endothelial cells, in which pro-inflammatory cytokines stimulate increased THY1 expression.
Relative to the neonatal and developing brain, the adult brain expresses far greater levels of THY1. Moreover, postnatal increase in Thy-1 expression coincides strongly with histological and physiological indicators of brain maturation. Thus, expression of Thy-1 is developmentally regulated in the brain. As the brain develops, expression of Thy1 mRNA precedes detection of protein by several days, thereby suggesting post-transcription regulation of Thy-1 mRNA as a mechanism for controlling temporal expression of Thy-1 protein in the developing brain. This mode of regulation has been shown to be an intrinsic attribute of immature neurons. Mature Thy1.1-expressing neurons fused with immature Thy1.2-negative neurons to form heterokaryons become Thy1 negative within 16 h. As factors that maintained the immature condition of the Thy1.2 negative neuron counterpart presumably give way, the heterokaryons express both Thy1.1 and Thy1.2 within 3-4 days after becoming negative. This suggests that a developmentally regulated diffusible suppressor molecule inhibits translation of the Thy1 protein in immature neurons. This method of regulation is perhaps a means to forgo the time needed to transcribe Thy-1 mRNA and thereby prime the immature neuron for immediate expression of Thy-1 protein to coincide with maturation. In response to injury, Thy1 expression in mature neurons mimics that of a developing neuron. In young adult rats, Thy1 expression dramatically decreases in dorsal root ganglion neurons two days post a crush injury of the sciatic nerve reaching a low around day four. Thy1 expression gradually returns to pre-injury levels 1 week after the sciatic nerve crush and coincides with recovery of sensory function. Central nervous system neurons do not have the same potential to recover from traumatic injury as peripheral nervous system neurons do. Response to traumatic injury by central nervous system ganglion cells also differs with respect to Thy1 expression. Specifically, optic nerve crush results in cell loss due to apoptosis after 2 weeks. Prior to any cell loss, levels of Thy1 mRNA decrease over the course of 7 days with no change in the number of Thy1 expressing cells. This suggests the decrease in Thy1 mRNA is an injury response rather than a consequence of apoptosis. The optic ganglion cells of Bax knockout mice are resistant to cell death following optic nerve crush. Despite this, the same decrease in Thy1 mRNA is observed following optic nerve crush in Bax knockout mice. Additional means of optic nerve injury also cause a decrease in Thy1 mRNA levels including intravitreal injections of N-methyl-D-aspartate and induced elevated intraocular pressure.
A soluble variety of THY1 is detectable in serum, cerebral spinal fluid, wound fluid from venous leg ulcers, and the synovial fluid from joints in rheumatoid arthritis. Cultured lung fibroblasts shed THY1 into the media when treated with pro-inflammatory cyokines, such as IL-1beta and TNF-alpha. The opposite effect is elicited in endothelial cells, in which pro-inflammatory cytokines stimulate increased THY1 expression.
Relative to the neonatal and developing brain, the adult brain expresses far greater levels of THY1. Moreover, postnatal increase in Thy-1 expression coincides strongly with histological and physiological indicators of brain maturation. Thus, expression of Thy-1 is developmentally regulated in the brain. As the brain develops, expression of Thy1 mRNA precedes detection of protein by several days, thereby suggesting post-transcription regulation of Thy-1 mRNA as a mechanism for controlling temporal expression of Thy-1 protein in the developing brain. This mode of regulation has been shown to be an intrinsic attribute of immature neurons. Mature Thy1.1-expressing neurons fused with immature Thy1.2-negative neurons to form heterokaryons become Thy1 negative within 16 h. As factors that maintained the immature condition of the Thy1.2 negative neuron counterpart presumably give way, the heterokaryons express both Thy1.1 and Thy1.2 within 3-4 days after becoming negative. This suggests that a developmentally regulated diffusible suppressor molecule inhibits translation of the Thy1 protein in immature neurons. This method of regulation is perhaps a means to forgo the time needed to transcribe Thy-1 mRNA and thereby prime the immature neuron for immediate expression of Thy-1 protein to coincide with maturation. In response to injury, Thy1 expression in mature neurons mimics that of a developing neuron. In young adult rats, Thy1 expression dramatically decreases in dorsal root ganglion neurons two days post a crush injury of the sciatic nerve reaching a low around day four. Thy1 expression gradually returns to pre-injury levels 1 week after the sciatic nerve crush and coincides with recovery of sensory function. Central nervous system neurons do not have the same potential to recover from traumatic injury as peripheral nervous system neurons do. Response to traumatic injury by central nervous system ganglion cells also differs with respect to Thy1 expression. Specifically, optic nerve crush results in cell loss due to apoptosis after 2 weeks. Prior to any cell loss, levels of Thy1 mRNA decrease over the course of 7 days with no change in the number of Thy1 expressing cells. This suggests the decrease in Thy1 mRNA is an injury response rather than a consequence of apoptosis. The optic ganglion cells of Bax knockout mice are resistant to cell death following optic nerve crush. Despite this, the same decrease in Thy1 mRNA is observed following optic nerve crush in Bax knockout mice. Additional means of optic nerve injury also cause a decrease in Thy1 mRNA levels including intravitreal injections of N-methyl-D-aspartate and induced elevated intraocular pressure.
Localisation
At the cell surface, Thy-1, like many GPI anchored proteins, localizes to cholesterol-rich lipid rafts. Release of Thy-1 into the extracellular space and into body fluids, such as serum and cerebral spinal fluid, occurs via unknown mechanisms. In-vitro, both mammalian GPI-PLD and bacterial PLC are capable of separating the diacyl glycerol from the remainder of the GPI-moiety. When Thy-1 is released from the cell surface by GPI-PLD the phosphate is not retained, but is when released by PLC. In order for Thy-1 to be susceptible to release by GPI-PLD, lipid rafts must be disrupted by detergents or saponins. PLC has no such requirement. However, Thy1 in certain cell types is more resistant to PLC. Thus, Thy-1 is thought to be afforded some protection from GPI-PLD catalyzed release by its localization within lipid rafts.
For Thy-1 to signal, it must be localized to its native lipid raft microdomain. Thy1 (-) neurons grown on a monolayer of astrocytes and expressing exogenous human THY1 or mouse Thy1.2 experienced inhibited neurite outgrowth. Yet under the same conditions, Thy1 (-) neurons expressing a construct of Thy1 in which the GPI anchor is replaced by the transmembrane domain of CD8 have normal neurite outgrowth. The transmembrane domain of CD8 does not localize Thy1 to lipid raft microdomains. This same construct was later used in experiments to assess the role of the Thy1 GPI anchor in modulating fibroblast phenotypes. Not unlike the results in the neuron experiments, Thy1 (-) fibroblasts made to express the Thy1-CD8 construct maintained their "negative" phenotype in that they remained insensitive to thrombospondin-mediated transient phosphorylation of FAK and SFK, focal adhesion disassembly, and migration.
For Thy-1 to signal, it must be localized to its native lipid raft microdomain. Thy1 (-) neurons grown on a monolayer of astrocytes and expressing exogenous human THY1 or mouse Thy1.2 experienced inhibited neurite outgrowth. Yet under the same conditions, Thy1 (-) neurons expressing a construct of Thy1 in which the GPI anchor is replaced by the transmembrane domain of CD8 have normal neurite outgrowth. The transmembrane domain of CD8 does not localize Thy1 to lipid raft microdomains. This same construct was later used in experiments to assess the role of the Thy1 GPI anchor in modulating fibroblast phenotypes. Not unlike the results in the neuron experiments, Thy1 (-) fibroblasts made to express the Thy1-CD8 construct maintained their "negative" phenotype in that they remained insensitive to thrombospondin-mediated transient phosphorylation of FAK and SFK, focal adhesion disassembly, and migration.
Function
The Thy1 antigen was initially discovered in an attempt to raise antiserum against leukemia-specific antigens from the CH3 mouse strain in the AKR mouse strain and vice versa. The antibodies were found to strongly label thymocytes as well as peripheral T cells, hence the name Thy1. Thy-1 has several immunological functions, most mediated through interactions with integrins. In particular, Thy1 binds integrins alphaMbeta2 and alphaXbeta2 which are both expressed by leukocytes. In cell adhesion assays, monocytes and polymorphonuclear cells adhere to exogenous Thy1-expressing CHO cells and activated THY1-expressing Human Dermal Microvascular Endothelial Cells (HDMECs). This adherence was found to be mediated by the interaction between THY1 and alphaMbeta2. Antibodies against alphaMbeta2 blocked adherence, while exogenously expressing alphaMbeta2 but not control CHO cells are able to retain biotynilated purified THY1 at their cell surface. Moreover, the interaction of THY1 with alphaMbeta2 was found to be vital in transendothelial migration of the aforementioned leukocytes across a monolayer of HDMECs. Thereby, THY1 is implicated in the regulation of leukocyte recruitment to sites of inflammation.
Early evidence showed surface Thy1 expression diminishes as thymocytes mature into T cells, suggesting a role for Thy-1 in regulating thymocyte lineage. Thymocytes from Thy1 null mice have a reduced maturation rate from the immature CD4+CD8+ double-positive to the CD4+ or CD8+ single-positive mature T cell. Thy1 supports adhesion of thymocytes to thymic epithelial cells. Accordingly, the contacts between thymus cells of the Thy1 null mice are atypical as evaluated by electron microscopy. The role of Thy-1 in thymocyte adhesion and maturation may not be mutually exclusive. Thy1 seems to have a continued role in T cell activity beyond maturation from thymocytes. Cross-linking Thy1 using bivalent antibodies against Thy1 results in T cell activation as indicated by proliferation and IL-2 synthesis. It is important to note there is no known ligand for Thy-1 that the cross-linking antibody presumably mimics. Traditionally, activation of a T cell is thought to require two simultaneous signals. One is a B7 family member engaging T cell CD28 receptor and the other is the T cell receptor being presented its specific antigen by an MHC. Cross-linking Thy1 with antibodies partially supplies the latter signal to activate T cells when the CD28 receptor is engaged by anti-CD28 antibodies. The activation is only partial because, although the T cells adhere to target cells and express perforin, granzyme B, and Fas ligand, they are unable to kill target cells. The specific role Thy1 has in T cell activation and thymocyte maturation in vivo, and whether they are related is unknown.
Thy-1 expression modulates fibroblast phenotype. Pulmonary fibroblasts sorted into Thy-1 (+) and (-) populations have dissimilar potential for differentiating into myofibroblasts, response to pro-inflammatory cytokines, and localization into areas of active fibrosis. Rat Thy1 (-) pulmonary fibroblasts have greater myofibroblastic differentiation relative to Thy1 (+) as assessed by contractility and myogenic gene expression of MyoD, myocardin, myf5, and myogenin, both at baseline and in response to fibrogenic mediators. In addition, Thy1 (-) fibroblasts resist apoptosis in a contracting collagen matrix. Thy1 (+) and (-) pulmonary fibroblasts have dissimilar production of and/or responses to various cytokines. In response to PDGF-BB, both populations undergo concentration-dependent proliferation. However, only the Thy1 (-) population proliferates in response to PDGF-AA. Consistent with the proliferation assays, both populations express PDGFR-beta. Only Thy1 (-) pulmonary fibroblast express PDGFR-alpha. Thrombospondin-1 or its N-terminal heparin-binding domain alone is a potent inducer of cell migration. Coordinated focal adhesion disassembly is critical for cell migration to occur. Thy1 (+) but not Thy1 (-) pulmonary fibroblasts respond to Thrombospondin-1/HEP-I with transient phosphorylation of FAK and SFK, focal adhesion disassembly, and migration. Exogenous expression of WT Thy1 by Thy1 (-) pulmonary fibroblasts creates sensitivity to Thrombospondin-1/HEP-I. It is important to note that as for other described functions of Thy-1, localization of Thy-1 within lipid rafts is required (see Localisation, above). Thy1 (-) pulmonary fibroblasts, in contrast to Thy1 (+), are capable of activating latent TGF-beta. Only Thy1 (-) murine pulmonary fibroblasts produce IL-1 in response to TNF-alpha. Moreover, proliferation and IL-6 expression induced by IL-1beta treatment is greater in Thy1 (-) cells relative to Thy1 (+). Phenotypes attributed to the Thy1 (-) and Thy1 (+) fibroblast population of one tissue may be different or reversed in those from another tissue. Unlike in the lung, only THY1 (+) orbital fibroblasts appear capable of differentiating into myofibroblasts, whereas THY1 (-) are incapable of doing so but are unique in their ability to differentiate into mature adipocytes. Thus, the effects of THY1 on cell signaling are likely to be context-dependent.
Purified Thy1 immobilized on microbeads has been shown to bind integrin beta3 on astrocytes causing them to form focal adhesion sites. However, inhibition of neurite outgrowth in Thy1 expressing neurons is not believed to require induced focal adhesion formation or factors emanating from the astrocytes. Thy1 (-) neurons expressing a construct of Thy1 at the cell surface that does not localize to lipid rafts and grown on a monolayer of astrocytes are capable of normal neurite outgrowth. Despite Thy1 being able to engage integrin beta3 on the astrocyte, neurite outgrowth is not inhibited. Thy1 requires correct localization to its native membrane micro domain to exert an inhibitory effect which suggests that Thy1 functions as a receptor of the neuron rather than a ligand for the astrocyte. Remarkably, Thy1 deficient mice have only subtle nervous system irregularities, including inhibition of hippocampal long-term potentiation in the dentate gyrus and failure to transmit social cues regarding food selection.
Early evidence showed surface Thy1 expression diminishes as thymocytes mature into T cells, suggesting a role for Thy-1 in regulating thymocyte lineage. Thymocytes from Thy1 null mice have a reduced maturation rate from the immature CD4+CD8+ double-positive to the CD4+ or CD8+ single-positive mature T cell. Thy1 supports adhesion of thymocytes to thymic epithelial cells. Accordingly, the contacts between thymus cells of the Thy1 null mice are atypical as evaluated by electron microscopy. The role of Thy-1 in thymocyte adhesion and maturation may not be mutually exclusive. Thy1 seems to have a continued role in T cell activity beyond maturation from thymocytes. Cross-linking Thy1 using bivalent antibodies against Thy1 results in T cell activation as indicated by proliferation and IL-2 synthesis. It is important to note there is no known ligand for Thy-1 that the cross-linking antibody presumably mimics. Traditionally, activation of a T cell is thought to require two simultaneous signals. One is a B7 family member engaging T cell CD28 receptor and the other is the T cell receptor being presented its specific antigen by an MHC. Cross-linking Thy1 with antibodies partially supplies the latter signal to activate T cells when the CD28 receptor is engaged by anti-CD28 antibodies. The activation is only partial because, although the T cells adhere to target cells and express perforin, granzyme B, and Fas ligand, they are unable to kill target cells. The specific role Thy1 has in T cell activation and thymocyte maturation in vivo, and whether they are related is unknown.
Thy-1 expression modulates fibroblast phenotype. Pulmonary fibroblasts sorted into Thy-1 (+) and (-) populations have dissimilar potential for differentiating into myofibroblasts, response to pro-inflammatory cytokines, and localization into areas of active fibrosis. Rat Thy1 (-) pulmonary fibroblasts have greater myofibroblastic differentiation relative to Thy1 (+) as assessed by contractility and myogenic gene expression of MyoD, myocardin, myf5, and myogenin, both at baseline and in response to fibrogenic mediators. In addition, Thy1 (-) fibroblasts resist apoptosis in a contracting collagen matrix. Thy1 (+) and (-) pulmonary fibroblasts have dissimilar production of and/or responses to various cytokines. In response to PDGF-BB, both populations undergo concentration-dependent proliferation. However, only the Thy1 (-) population proliferates in response to PDGF-AA. Consistent with the proliferation assays, both populations express PDGFR-beta. Only Thy1 (-) pulmonary fibroblast express PDGFR-alpha. Thrombospondin-1 or its N-terminal heparin-binding domain alone is a potent inducer of cell migration. Coordinated focal adhesion disassembly is critical for cell migration to occur. Thy1 (+) but not Thy1 (-) pulmonary fibroblasts respond to Thrombospondin-1/HEP-I with transient phosphorylation of FAK and SFK, focal adhesion disassembly, and migration. Exogenous expression of WT Thy1 by Thy1 (-) pulmonary fibroblasts creates sensitivity to Thrombospondin-1/HEP-I. It is important to note that as for other described functions of Thy-1, localization of Thy-1 within lipid rafts is required (see Localisation, above). Thy1 (-) pulmonary fibroblasts, in contrast to Thy1 (+), are capable of activating latent TGF-beta. Only Thy1 (-) murine pulmonary fibroblasts produce IL-1 in response to TNF-alpha. Moreover, proliferation and IL-6 expression induced by IL-1beta treatment is greater in Thy1 (-) cells relative to Thy1 (+). Phenotypes attributed to the Thy1 (-) and Thy1 (+) fibroblast population of one tissue may be different or reversed in those from another tissue. Unlike in the lung, only THY1 (+) orbital fibroblasts appear capable of differentiating into myofibroblasts, whereas THY1 (-) are incapable of doing so but are unique in their ability to differentiate into mature adipocytes. Thus, the effects of THY1 on cell signaling are likely to be context-dependent.
Purified Thy1 immobilized on microbeads has been shown to bind integrin beta3 on astrocytes causing them to form focal adhesion sites. However, inhibition of neurite outgrowth in Thy1 expressing neurons is not believed to require induced focal adhesion formation or factors emanating from the astrocytes. Thy1 (-) neurons expressing a construct of Thy1 at the cell surface that does not localize to lipid rafts and grown on a monolayer of astrocytes are capable of normal neurite outgrowth. Despite Thy1 being able to engage integrin beta3 on the astrocyte, neurite outgrowth is not inhibited. Thy1 requires correct localization to its native membrane micro domain to exert an inhibitory effect which suggests that Thy1 functions as a receptor of the neuron rather than a ligand for the astrocyte. Remarkably, Thy1 deficient mice have only subtle nervous system irregularities, including inhibition of hippocampal long-term potentiation in the dentate gyrus and failure to transmit social cues regarding food selection.
Implicated in
Entity name
Nasopharyngeal carcinoma
Note
In nasopharyngeal carcinoma, THY1 is believed to function as a tumor suppressor. The tumorigenic NPC cell line, HONE1, decrease colony formation with addition of an intact chromosome 11 via microcell-mediated transfer and coincides with reexpression of THY-1. The THY1 gene is located within region 11q22-23 which is critical for tumorigenicity in NPC. Additionally, tumor segregants of the HONE1 microcell hybrids were all negative for THY1. THY1 expression is decreased in more invasive/metastatic NPC and this is thought to occur via epigenetic silencing.
Entity name
Ovarian cancer
Note
The loss of heterozygosity at 11q23.3-q24.3 for patients with ovarian cancer is associated with poor prognosis. This region of chromosome 11 contains the THY1 gene. THY1 expression in the tumorigenic ovarian cancer cell line, SKOV-3, induced by either microcell-mediated chromosome 11 transfer or THY1 expression inducible system suppresses tumorigenicity.
Entity name
Idiopathic pulmonary fibrosis (IPF)
Note
Evidence from both in vivo and in vitro experiments implicates loss of fibroblast THY1 is important in the disease Idiopathic Pulmonary Fibrosis (IPF). The most characteristic histopathologic feature of IPF is aggregates of proliferating fibroblasts and myofibroblasts called fibrotic foci. The number of FF directly correlates with severity of the disease. Though the vast majority of quiescent lung fibroblasts are THY1 positive, FF are exclusively occupied by THY1 (-) myofibroblasts. Thy1 (-) lung fibroblasts have a more fibrotic phenotype including response to profibrotic cytokines and propensity to differentiate into myofibroblasts. Moreover, the chemically induced model for lung fibrosis, intra-tracheal administered bleomycin, is more severe in Thy1 knockout mice with respect to accumulation of myofibroblasts, collagen, and increased activation of TGF-beta. THY1 expression has been shown to be epigenetically silenced by DNA hypermethylation in fibroblasts from IPF lesions in vivo and in vitro.
Prognosis
The prognosis for a patient with IPF is almost universally poor, with a mean survival of only 2 to 4 years after diagnosis. Onset of the disease and subsequent diagnosis usually occur after the age of 50. Over the course of the disease, patients suffer severe dyspnea. For the estimated 40000 to 130000 IPF patients in the United States, there is no medical intervention that affords a survival benefit save for a lung transplant.
Entity name
Experimental glomerulonephritis
Note
Intravenous administration of crosslinking anti-Thy1 antibodies to rats induces glumerulonephritis and serves as a model for study of the disease. The use of Thy1 antibodies for this purpose is supported by in vitro experiments. Cross linking Thy1 expressed on glomerular mesangial cells with anti-Thy1 antibodies induces apoptosis as confirmed by TdT-mediated dUTP nick-end labeling (TUNEL) and annexin V assays. Glumerulonephritis induced with anti-Thy1 antibodies in vivo has recently been shown to result from the combination of complement-mediated necrosis as well as apoptosis.
Entity name
Gravesophthalmopathy (GO)
Note
GO is characterized by an increase in volume of the extraocular muscles and/or the intraorbital adipose tissues which causes the eyeball to bulge from the orbit. Intraorbital fibroblasts, including those that reside around and within the extraocular muscles, have a pathogenic role in this disease. The THY1 (+), but not the THY1 (-), orbital fibroblast subpopulation differentiates into myofibroblasts as indicated by alpha-smooth muscle actin expression. The opposite is true for differentiation into lipofibroblasts assessed by accumulation of cytoplasmic lipid droplets. The orbital adipose/connective tissue taken from GO patients was shown to have greater THY1 mRNA and protein expression relative to the same tissue retrieved from individuals with no history of Gravesdisease whose corneas were being procured for transplantation. Fibroblasts cultured from these two tissue sources were examined for THY1 expression. As with total tissue levels, there was greater expression of THY1 by fibroblasts cultured from tissue obtained from GO patients.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 6971158 | 1981 | Ontogeny of murine T lymphocytes. I. Maturation of thymocytes induced in vitro by tumor necrosis factor-positive serum (TNF+)1,2. | Abbott J et al |
| 15104276 | 2004 | The role of the THY1 gene in human ovarian cancer suppression based on transfection studies. | Abeysinghe HR et al |
| 2900838 | 1988 | Characterization of a hydrophilic form of Thy-1 purified from human cerebrospinal fluid. | Almqvist P et al |
| 62283 | 1976 | Chemical characterisation of the Thy-1 glycoproteins from the membranes of rat thymocytes and brain. | Barclay AN et al |
| 372497 | 1979 | Localization of the Thy-1 antigen in the cerebellar cortex of rat brain by immunofluorescence during postnatal development. | Barclay AN et al |
| 15033989 | 2004 | Thrombospondin-1-induced focal adhesion disassembly in fibroblasts requires Thy-1 surface expression, lipid raft integrity, and Src activation. | Barker TH et al |
| 7908190 | 1994 | Saponin-induced release of cell-surface-anchored Thy-1 by serum glycosylphosphatidylinositol-specific phospholipase D. | Bergman AS et al |
| 19422052 | 2009 | Roles and regulation of Thy-1, a context-dependent modulator of cell phenotype. | Bradley JE et al |
| 12112467 | 2002 | Characterization of transgene expression and Cre recombinase activity in a panel of Thy-1 promoter-Cre transgenic mice. | Campsall KD et al |
| 15547945 | 2005 | Role of Thy-1 in in vivo and in vitro neural development and regeneration of dorsal root ganglionic neurons. | Chen CH et al |
| 15850796 | 2005 | Characterization of alphaX I-domain binding to Thy-1. | Choi J et al |
| 10561463 | 1999 | The glycan processing and site occupancy of recombinant Thy-1 is markedly affected by the presence of a glycosylphosphatidylinositol anchor. | Devasahayam M et al |
| 2866091 | 1985 | Structure of the murine Thy-1 gene. | Giguére V et al |
| 6142077 | 1984 | T cell-activating properties of an anti-Thy-1 monoclonal antibody. Possible analogy to OKT3/Leu-4. | Gunter KC et al |
| 15356100 | 2004 | Thy-1: more than a mouse pan-T cell marker. | Haeryfar SM et al |
| 10409250 | 1999 | Differential expression of platelet-derived growth factor-alpha receptor by Thy-1(-) and Thy-1(+) lung fibroblasts. | Hagood JS et al |
| 16049324 | 2005 | Loss of fibroblast Thy-1 expression correlates with lung fibrogenesis. | Hagood JS et al |
| 1671083 | 1991 | Thy-1 supports adhesion of mouse thymocytes to thymic epithelial cells through a Ca2(+)-independent mechanism. | He HT et al |
| 6965519 | 1980 | T-lymphocyte differentiation is accompanied by increase in sialic acid content of Thy-1 antigen. | Hoessli D et al |
| 9285719 | 1997 | Thymocytes in Thy-1-/- mice show augmented TCR signaling and impaired differentiation. | Hueber AO et al |
| 9056477 | 1997 | Interleukin-1alpha regulates Thy-1 expression on rat vascular endothelial cells. | Ishizu A et al |
| 18976167 | 2008 | Evidence for enhanced Thy-1 (CD90) expression in orbital fibroblasts of patients with Graves' ophthalmopathy. | Khoo TK et al |
| 9382830 | 1997 | T-cell regulation: Thy-1 - hiding in full view. | Killeen N et al |
| 14507638 | 2003 | Thy-1 expression in human fibroblast subsets defines myofibroblastic or lipofibroblastic phenotypes. | Koumas L et al |
| 19661226 | 2010 | Perimysial fibroblasts of extraocular muscle, as unique as the muscle fibers. | Kusner LL et al |
| 11470407 | 2001 | Thy-1 binds to integrin beta(3) on astrocytes and triggers formation of focal contact sites. | Leyton L et al |
| 2865681 | 1985 | Phosphatidylinositol is the membrane-anchoring domain of the Thy-1 glycoprotein. | Low MG et al |
| 16007174 | 2005 | THY1 is a candidate tumour suppressor gene with decreased expression in metastatic nasopharyngeal carcinoma. | Lung HL et al |
| 10662668 | 2000 | Mice lacking the cell adhesion molecule Thy-1 fail to use socially transmitted cues to direct their choice of food. | Mayeux-Portas V et al |
| 7462633 | 1981 | Human thy-1: unusual localization and possible functional significance in lymphoid tissues. | McKenzie JL et al |
| 8773358 | 1996 | Thy-1 antigen mediates apoptosis of rat glomerular cells in vitro and in vivo. | Morita H et al |
| 8587606 | 1996 | Normal spatial learning despite regional inhibition of LTP in mice lacking Thy-1. | Nosten-Bertrand M et al |
| 1976521 | 1990 | Differential expression of interleukin 1 alpha by Thy-1+ and Thy-1- lung fibroblast subpopulations: enhancement of interleukin 1 alpha production by tumor necrosis factor-alpha. | Phipps RP et al |
| 14207060 | 1964 | THE AKR THYMIC ANTIGEN AND ITS DISTRIBUTION IN LEUKEMIAS AND NERVOUS TISSUES. | REIF AE et al |
| 16996153 | 2006 | Thy-1, a versatile modulator of signaling affecting cellular adhesion, proliferation, survival, and cytokine/growth factor responses. | Rege TA et al |
| 17027000 | 2006 | Thy-1, via its GPI anchor, modulates Src family kinase and focal adhesion kinase phosphorylation and subcellular localization, and fibroblast migration, in response to thrombospondin-1/hep I. | Rege TA et al |
| 11069627 | 2000 | A ligand of human thy-1 is localized on polymorphonuclear leukocytes and monocytes and mediates the binding to activated thy-1-positive microvascular endothelial cells and fibroblasts. | Saalbach A et al |
| 2570164 | 1989 | Evidence from neuronal heterokaryons for a trans-acting factor suppressing Thy-1 expression during neuronal development. | Saleh M et al |
| 18556592 | 2008 | Thy-1 promoter hypermethylation: a novel epigenetic pathogenic mechanism in pulmonary fibrosis. | Sanders YY et al |
| 10607429 | 1999 | T-cell-activating monoclonal antibodies, reacting with both leukocytes and erythrocytes, recognize the guinea pig Thy-1 differentiation antigen: characterization and cloning of guinea pig CD90. | Schäfer H et al |
| 11509915 | 2001 | Changes in Thy1 gene expression associated with damaged retinal ganglion cells. | Schlamp CL et al |
| 5783714 | 1969 | Antigenic changes in lymph-node cells after administration of antiserum to thymus cells. | Schlesinger M et al |
| 17934117 | 2007 | Molecular targets in pulmonary fibrosis: the myofibroblast in focus. | Scotton CJ et al |
| 6172518 | 1982 | Definition of a Thy-1 determinant on human neuroblastoma, glioma, sarcoma, and teratoma cells with a monoclonal antibody. | Seeger RC et al |
| 2864690 | 1985 | The human Thy-1 gene: structure and chromosomal location. | Seki T et al |
| 11014985 | 2000 | Complement-mediated killing of mesangial cells in experimental glomerulonephritis: cell death by a combination of apoptosis and necrosis. | Shimizu A et al |
| 2906111 | 1988 | Transcriptional unit of the murine Thy-1 gene: different distribution of transcription initiation sites in brain. | Spanopoulou E et al |
| 17387221 | 2007 | FcepsilonRI and Thy-1 domains have unique protein and lipid compositions. | Surviladze Z et al |
| 7983148 | 1994 | The mode of anchorage to the cell surface determines both the function and the membrane location of Thy-1 glycoprotein. | Tiveron MC et al |
| 9683268 | 1997 | A molecular basis for species differences in Thy-1 expression patterns. | Tokugawa Y et al |
| 1968831 | 1990 | Tissue-specific control elements of the Thy-1 gene. | Vidal M et al |
| 15004192 | 2004 | Human Thy-1 (CD90) on activated endothelial cells is a counterreceptor for the leukocyte integrin Mac-1 (CD11b/CD18). | Wetzel A et al |
| 8104555 | 1993 | Comparative analysis of the N-glycans of rat, mouse and human Thy-1. Site-specific oligosaccharide patterns of neural Thy-1, a member of the immunoglobulin superfamily. | Williams AF et al |
| 1345938 | 1992 | Expression of the neuronal surface glycoprotein Thy-1 does not follow appearance of its mRNA in developing mouse Purkinje cells. | Xue GP et al |
| 1685113 | 1991 | The surface glycoprotein Thy-1 is excluded from growing axons during development: a study of the expression of Thy-1 during axogenesis in hippocampus and hindbrain. | Xue GP et al |
| 12759414 | 2003 | NK cell tolerance in mixed allogeneic chimeras. | Zhao Y et al |
| 15277239 | 2004 | Thy-1 expression regulates the ability of rat lung fibroblasts to activate transforming growth factor-beta in response to fibrogenic stimuli. | Zhou Y et al |
Other Information
Locus ID:
NCBI: 7070
MIM: 188230
HGNC: 11801
Ensembl: ENSG00000154096
Variants:
dbSNP: 7070
ClinVar: 7070
TCGA: ENSG00000154096
COSMIC: THY1
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 36633486 | 2023 | Integrative analysis implicates the significance of m6A in the liver fibrosis of biliary atresia by regulating THY1. | 2 |
| 36949573 | 2023 | CD44, CD90 and CD96 expression in immune thrombocytopenia purpura (ITP) patients. | 0 |
| 38254918 | 2023 | High Expression of THY1 in Intestinal Gastric Cancer as a Key Factor in Tumor Biology: A Poor Prognosis-Independent Marker Related to the Epithelial-Mesenchymal Transition Profile. | 0 |
| 36633486 | 2023 | Integrative analysis implicates the significance of m6A in the liver fibrosis of biliary atresia by regulating THY1. | 2 |
| 36949573 | 2023 | CD44, CD90 and CD96 expression in immune thrombocytopenia purpura (ITP) patients. | 0 |
| 38254918 | 2023 | High Expression of THY1 in Intestinal Gastric Cancer as a Key Factor in Tumor Biology: A Poor Prognosis-Independent Marker Related to the Epithelial-Mesenchymal Transition Profile. | 0 |
| 34737243 | 2022 | Circulating Cancer Stem Cells Expressing EpCAM/CD90 in Hepatocellular Carcinoma: A Pilot Study for Predicting Tumor Recurrence after Living Donor Liver Transplantation. | 6 |
| 34737243 | 2022 | Circulating Cancer Stem Cells Expressing EpCAM/CD90 in Hepatocellular Carcinoma: A Pilot Study for Predicting Tumor Recurrence after Living Donor Liver Transplantation. | 6 |
| 33242496 | 2021 | X-aptamers targeting Thy-1 membrane glycoprotein in pancreatic ductal adenocarcinoma. | 3 |
| 34230598 | 2021 | Innovative finding of 266-nm laser regulating CD90 levels in SDSCs. | 0 |
| 33242496 | 2021 | X-aptamers targeting Thy-1 membrane glycoprotein in pancreatic ductal adenocarcinoma. | 3 |
| 34230598 | 2021 | Innovative finding of 266-nm laser regulating CD90 levels in SDSCs. | 0 |
| 32396525 | 2020 | Transcription factor YY1 inhibits the expression of THY1 to promote interstitial pulmonary fibrosis by activating the HSF1/miR-214 axis. | 6 |
| 33114405 | 2020 | CD90 Is Dispensable for White and Beige/Brown Adipocyte Differentiation. | 0 |
| 33322687 | 2020 | The mRNA Distribution of Cancer Stem Cell Marker CD90/Thy-1 Is Comparable in Hepatocellular Carcinoma of Eastern and Western Populations. | 3 |
Citation
John E Bradley ; James S Hagood
THY1 (Thy-1 cell surface antigen)
Atlas Genet Cytogenet Oncol Haematol. 2010-01-01
Online version: http://atlasgeneticsoncology.org/gene/45672/thy1
