PRKCI (protein kinase C, iota)
2012-06-01 Verline Justilien , Alan P Fields AffiliationDepartment of Cancer Biology, Mayo Clinic College of Medicine, Jacksonville, Florida, 32224 USA
Identity
HGNC
LOCATION
3q26.2
IMAGE

LEGEND
Location sequence of PRKCI on Chromosome 3. PRKCI gene is indicated by red arrow.
LOCUSID
ALIAS
DXS1179E,PKCI,nPKC-iota
FUSION GENES
DNA/RNA
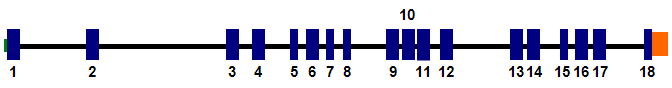
Exon-intron structure of the PRKCI gene. Blue vertical bars correspond to exons, green bar represents 5UTR and orange 3UTR.
Description
The PRKCI gene is composed of 18 exons and spans 83618 bases on the plus strand.
Transcription
The PRKCI transcript (NM_002740) contains 4884 bases and the open reading frame spans from 239 to 2029.
Pseudogene
There is a single exon pseudogene mapped on chromosome X.
Proteins
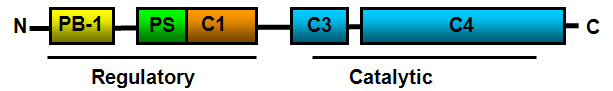
Schematic diagram showing the domain structure of PKCι. PB-1 Phox-Bem1; PS: auto-inhibitory pseudosubstrate sequence.
Description
PKCι consist of 596 amino acids and has a molecular mass of 68262 Da. PKCι is a member of the PKCs, a diverse family of lipid dependent serine/threonine kinases. PKCι activity can be regulated by lipid second messengers (ceramide, phosphatidylinositol 3,4,5-P3, and phosphatidic acid), phosphoinositide-dependent kinase (PDK1), tyrosine phosphorylation and specific protein-protein interactions. The PB1 domain within the N-terminal regulatory domain mediates protein-protein interactions between PKCι and other PB1 domain containing proteins such as ZIP/p62 (Hirano et al., 2004; Puls et al., 1997), Par-6 (partitioning-defective 6) (Joberty et al., 2000; Lin et al., 2000; Noda et al., 2001; Qiu et al., 2000) and MEK5 (MAPK (mitogen-activated protein kinase)/ERK (extracellular-signal-regulated kinase) kinase 5) (Diaz-Meco and Moscat, 2001; Hirano et al., 2004). In the inactive state, the PKCι PS is positioned in the substrate binding cavity in the kinase domain and is displaced upon PKCι activation. Phosphatidylserine binds the C1 domain to anchor PKCι to the membrane. The PKCι catalytic domain is subdivided into the C3 and C4 domains that mediate ATP-binding and substrate binding.
Expression
PKCι is widely expressed with varying levels in different tissues (Selbie et al., 1993).
Localisation
PKCι is mainly expressed in the cytoplasm. PKCι is translocated to the cell membrane in response to second messengers and colocalizes with p62/ZIP in lysosome-targeted endosomes (Sanchez et al., 1998). Src phosphorylation leads to translocation of PKCι into the nucleus (White et al., 2002) where it forms a complex with Cdk7 (Win and Acevedo-Duncan, 2008).
Function
PKCι is a lipid-dependent, serine/threonine kinase. PKCι participates a number of signaling pathways that regulate cell survival (Sanz et al., 1999; Wooten et al., 1999; Xie et al., 2000), differentiation (Wooten et al., 2000), polarity (Joberty et al., 2000), and microtubule dynamics in the early secretory pathway (Tisdale, 2002).
Homology
PRKCI is highly evolutionarily conserved. PKCι and PKCζ exhibit 72% overall amino acid sequence homology and 86% identity within the kinase domain. PKCι shows less homology with the other PKC isoform, with less than 53% identity in the highly conserved catalytic domain (Selbie et al., 1993).
Mutations
Germinal
No germline mutations in the PRKCI gene have been reported.
Somatic
The PKCι gene is amplified as part of the 3q26 amplicon in lung (Regala et al., 2005b), esophageal (Yang et al., 2008) and ovarian (Eder et al., 2005; Zhang et al., 2006) cancers. A P118L mutation was found in a metastatic melanoma sample (Greenman et al., 2007).
Implicated in
Entity name
Various cancers
Note
PKCι overexpression has been observed in numerous human cancers including cancers of the lung (Regala et al., 2005b), pancreas (Scotti et al., 2010), stomach (Takagawa et al., 2010), colon (Murray et al., 2004), esophagus (Yang et al., 2008), liver (Du et al., 2009), bile duct (Li et al., 2008), breast (Kojima et al., 2008), ovary (Weichert et al., 2003; Eder et al., 2005; Zhang et al., 2006), prostate (Ishiguro et al., 2009), and brain (Patel et al., 2008). PKCι is itself an oncogene, which appears to be activated through tumor-specific overexpression. In addition, however, PKCι is activated downstream of other oncogenes including oncogenic Ras, Bcr-Abl and Src.
Entity name
Non Small Cell Lung Cancer (NSCLC)
Prognosis
Elevated levels of PKCι expression correlate with poor clinical outcome in NSCLC patients (Regala et al., 2005b).
Cytogenetics
The PRKCI gene is amplified as part of the 3q26 amplicon in NSCLC.
Oncogenesis
PKCι is an oncogene in NSCLC. PRKCI is amplified as part of the 3q26 amplicon in NSCLC and amplication drives PKCι overexpression in NSCLC cell lines and primary NSCLC tumours. PKCι is required for transformed (anchorage-independent) growth and invasion of human NSCLC cells (Frederick et al., 2008; Regala et al., 2005a). Disruption of the Prkci gene inhibits oncogenic Kras induced expansion and transformation of tumor-initiating, lung stem-like cells. Consequently, genetic loss of Prkci dramatically inhibits Kras-initiated hyperplasia and subsequent lung tumor formation in vivo. PKCι enhances resistance of NSCLC to NNK-induced apoptosis by phosphorylating the pro-apoptotic protein BAD (Jin et al., 2005). PKCι forms an oncogeneic complex with Par6 that activates a Rac1-Mek-Erk signaling axis that drives the transformed growth and invasion of NSCLC cells in vitro (Frederick et al., 2008; Regala et al., 2005a) and tumorigenicity in vivo (Regala et al., 2005a). PKCι and the oncogene ECT2 are genetically linked through coordinate gene amplification as part of the 3q26 amplicon in NSCLC tumors (Justilien and Fields, 2009). PKCι phosphorylates Ect2 and forms an oncogenic PKCι-Par6-Ect2 complex that drives NSCLC cell transformation by activating Rac1 (Justilien and Fields, 2009; Justilien et al., 2011). Expression of MMP10 is regulated through the PKCι-Par6-Rac1 signaling axis and MMP10 represents a key downstream effector in PKCι mediated transformation in lung cancer cells that is required for transformed growth and invasion (Frederick et al., 2008). PKCι also regulates expression of COPB2, ELF3, RFC4, and PLS1 in primary lung adenocarcinoma (Erdogan et al., 2009). The PKCι inhibitor aurothiomalate (ATM) disrupts the PB1-PB1 domain interaction between PKCι and Par6 and inhibits PKCι-mediated Rac1 activation and blocks anchorage-independent growth of NSCLC cells in vitro and tumorigenicity in vivo (Erdogan et al., 2006; Stallings-Mann, 2006).
Entity name
Colon cancer
Oncogenesis
PKCι expression is elevated in human colon tumors, AOM-induced colon tumors in mice (Murray et al., 2004) and intestinal tumors in APCMin/+ mice (Murray et al., 2009; Oster and Leitges, 2006). Expression of caPKCι in the colonic epithelium of mice led to an increase in the number of AOM-induced colon tumors, and promoted tumor progression from benign adenoma to malignant intramucosal carcinoma (Murray et al., 2004) PKCι is required for oncogenic Ras-mediated transformation of the intestinal epithelium in vitro and in vivo. PKCι is also required for the formation of intestinal tumors in APCMin/+ mice (Murray et al., 2009).
Entity name
Pancreatic cancer
Prognosis
PKCι overexpression predicts poor survival in pancreatic cancer patients (Scotti et al., 2010).
Oncogenesis
PKCι is significantly overexpressed in human pancreatic cancer. Knock down of PKCι expression using lentiviral-mediated shRNA blocked transformed (anchorage-independent) growth and invasion of human Pancreatic Ductal Adenocarcinoma (PDAC) cells (Scotti et al., 2010). Disruption of PKCι expression also blocks tumorigenicity of PDAC cell tumors injected orthotopically into the pancreas (Scotti et al., 2010). Analysis of human PDAC cells after orthotopic injection into the mouse pancreas revealed that PKCι-deficient tumor cells yielded significantly smaller tumors and significantly fewer metastases to the kidney, liver, diaphragm and mesentery (Scotti et al., 2010). The Rac1-MEK/ERK1/2 signaling axis is required for PKCiota-mediated transformed growth and cellular invasion of PDAC cells (Scotti et al., 2010).
Entity name
Ovarian cancer
Prognosis
PKCι expression is a strong predictor of survival when combined in a multi-variate analysis with tumor cyclin E expression (Eder et al., 2005).
Cytogenetics
The PRKCI gene is amplified as part of the 3q26 amplicon in ovarian cancer (Eder et al., 2005).
Oncogenesis
PKCι is frequently overexpressed in patients with ovarian cancer (Eder et al., 2005; Weichert et al., 2003; Zhang et al., 2006). PKCι expression in ovarian cancer patients correlates with tumor stage suggesting the involvement of PKCι in tumor progression and aggressiveness (Eder et al., 2005; Weichert et al., 2003; Zhang et al., 2006). Decreased PKCι expression reduced anchorage-independent growth of ovarian cancer cells, whereas overexpression of PKCι promoted murine ovarian surface epithelium transformation (Zhang et al., 2006).
Entity name
Oncogenesis
PKCι is highly expressed in human K562 leukemia cells and functions as a survival gene in chronic myelogenous leukemia (CML). The chimeric tyrosine kinase oncogene Bcr-Abl activates a Ras/Mek/Erk signaling pathway that stimulates PKCι expression through an Elk1 transcription factor site in the proximal promoter of PKCι (Gustafson et al., 2004). Bcr-Abl activation of PKCι is necessary and sufficient to mediate apoptotic resistance to chemotherapy in K562 CML cells (Murray and Fields, 1997).
Entity name
Gliomas
Oncogenesis
PKCι is overexpressed in glioblastoma multiforme. PKCι is required for survival and chemoresistance of glioblastoma cells. Genetic disruption of PKCι expression results in sensitization of glioblastoma cells to cisplatin (Baldwin et al., 2008). RNAi mediated depletion of PKCι also blocks the proliferative and invasive properties of glioma cell lines in vitro (Baldwin et al., 2008; Patel et al., 2008). PKCι promotes survival in glioblastoma cells through attenuation of p38 mitogen-activated protein kinase signaling that protects these cells against cytotoxicity to chemotherapeutic agents (Baldwin et al., 2008).
Entity name
Esophageal cancer
Cytogenetics
PRKCI gene is amplified as part of the 3q26 amplicon (Yang et al., 2008).
Oncogenesis
PRKCI is amplified in 53% of esophageal squamous cell carcinomas (ESCC) and PKCι protein expression correlated with PRKCI gene amplification in these tumors (Yang et al., 2008). Examination of clinicopathologic features of ESCC tumors revealed a significant correlation between PRKCI expression and larger tumor size, later stage and lymph node metastasis suggesting that PRKCI overexpression is a hallmark of tumor progression and metastasis in ESCC (Yang et al., 2008).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 18212741 | 2008 | Regulation of glioblastoma cell invasion by PKC iota and RhoB. | Baldwin RM et al |
| 11158308 | 2001 | MEK5, a new target of the atypical protein kinase C isoforms in mitogenic signaling. | Diaz-Meco MT et al |
| 19290490 | 2009 | Expression of P-aPKC-iota, E-cadherin, and beta-catenin related to invasion and metastasis in hepatocellular carcinoma. | Du GS et al |
| 16116079 | 2005 | Atypical PKCiota contributes to poor prognosis through loss of apical-basal polarity and cyclin E overexpression in ovarian cancer. | Eder AM et al |
| 19223491 | 2009 | Meta-analysis of oncogenic protein kinase Ciota signaling in lung adenocarcinoma. | Erdogan E et al |
| 16861740 | 2006 | Aurothiomalate inhibits transformed growth by targeting the PB1 domain of protein kinase Ciota. | Erdogan E et al |
| 18427549 | 2008 | Matrix metalloproteinase-10 is a critical effector of protein kinase Ciota-Par6alpha-mediated lung cancer. | Frederick LA et al |
| 17344846 | 2007 | Patterns of somatic mutation in human cancer genomes. | Greenman C et al |
| 14670960 | 2004 | Bcr-Abl regulates protein kinase Ciota (PKCiota) transcription via an Elk1 site in the PKCiota promoter. | Gustafson WC et al |
| 15143057 | 2004 | Solution structure of atypical protein kinase C PB1 domain and its mode of interaction with ZIP/p62 and MEK5. | Hirano Y et al |
| 19805306 | 2009 | aPKClambda/iota promotes growth of prostate cancer cells in an autocrine manner through transcriptional activation of interleukin-6. | Ishiguro H et al |
| 15705582 | 2005 | Survival function of protein kinase C{iota} as a novel nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-activated bad kinase. | Jin Z et al |
| 10934474 | 2000 | The cell-polarity protein Par6 links Par3 and atypical protein kinase C to Cdc42. | Joberty G et al |
| 21189248 | 2011 | Oncogenic activity of Ect2 is regulated through protein kinase C iota-mediated phosphorylation. | Justilien V et al |
| 18538170 | 2008 | The overexpression and altered localization of the atypical protein kinase C lambda/iota in breast cancer correlates with the pathologic type of these tumors. | Kojima Y et al |
| 18234642 | 2008 | Correlation of aPKC-iota and E-cadherin expression with invasion and prognosis of cholangiocarcinoma. | Li Q et al |
| 10934475 | 2000 | A mammalian PAR-3-PAR-6 complex implicated in Cdc42/Rac1 and aPKC signalling and cell polarity. | Lin D et al |
| 9346882 | 1997 | Atypical protein kinase C iota protects human leukemia cells against drug-induced apoptosis. | Murray NR et al |
| 15024028 | 2004 | Protein kinase Ciota is required for Ras transformation and colon carcinogenesis in vivo. | Murray NR et al |
| 19147581 | 2009 | Protein kinase C betaII and PKCiota/lambda: collaborating partners in colon cancer promotion and progression. | Murray NR et al |
| 11260256 | 2001 | Human homologues of the Caenorhabditis elegans cell polarity protein PAR6 as an adaptor that links the small GTPases Rac and Cdc42 to atypical protein kinase C. | Noda Y et al |
| 16849539 | 2006 | Protein kinase C alpha but not PKCzeta suppresses intestinal tumor formation in ApcMin/+ mice. | Oster H et al |
| 18211289 | 2008 | Involvement of PKC-iota in glioma proliferation. | Patel R et al |
| 9177193 | 1997 | Interaction of protein kinase C zeta with ZIP, a novel protein kinase C-binding protein. | Puls A et al |
| 10873802 | 2000 | A human homolog of the C. elegans polarity determinant Par-6 links Rac and Cdc42 to PKCzeta signaling and cell transformation. | Qiu RG et al |
| 16204062 | 2005 | Atypical protein kinase C iota is an oncogene in human non-small cell lung cancer. | Regala RP et al |
| 9566925 | 1998 | Localization of atypical protein kinase C isoforms into lysosome-targeted endosomes through interaction with p62. | Sanchez P et al |
| 10356400 | 1999 | The interaction of p62 with RIP links the atypical PKCs to NF-kappaB activation. | Sanz L et al |
| 20179210 | 2010 | Protein kinase Ciota is required for pancreatic cancer cell transformed growth and tumorigenesis. | Scotti ML et al |
| 8226978 | 1993 | Molecular cloning and characterization of PKC iota, an atypical isoform of protein kinase C derived from insulin-secreting cells. | Selbie LA et al |
| 16452237 | 2006 | A novel small-molecule inhibitor of protein kinase Ciota blocks transformed growth of non-small-cell lung cancer cells. | Stallings-Mann M et al |
| 19774416 | 2010 | High expression of atypical protein kinase C lambda/iota in gastric cancer as a prognostic factor for recurrence. | Takagawa R et al |
| 11724794 | 2002 | Glyceraldehyde-3-phosphate dehydrogenase is phosphorylated by protein kinase Ciota /lambda and plays a role in microtubule dynamics in the early secretory pathway. | Tisdale EJ et al |
| 12888898 | 2003 | Protein kinase C isoform expression in ovarian carcinoma correlates with indicators of poor prognosis. | Weichert W et al |
| 11891849 | 2002 | Phosphorylation of tyrosine 256 facilitates nuclear import of atypical protein kinase C. | White WO et al |
| 18571841 | 2008 | Atypical protein kinase C phosphorylates IKKalphabeta in transformed non-malignant and malignant prostate cell survival. | Win HY et al |
| 10848576 | 2000 | Mapping of atypical protein kinase C within the nerve growth factor signaling cascade: relationship to differentiation and survival of PC12 cells. | Wooten MW et al |
| 11042363 | 2000 | Protein kinase C iota protects neural cells against apoptosis induced by amyloid beta-peptide. | Xie J et al |
| 17990328 | 2008 | Amplification of PRKCI, located in 3q26, is associated with lymph node metastasis in esophageal squamous cell carcinoma. | Yang YL et al |
| 16651413 | 2006 | Integrative genomic analysis of protein kinase C (PKC) family identifies PKCiota as a biomarker and potential oncogene in ovarian carcinoma. | Zhang L et al |
Other Information
Locus ID:
NCBI: 5584
MIM: 600539
HGNC: 9404
Ensembl: ENSG00000163558
Variants:
dbSNP: 5584
ClinVar: 5584
TCGA: ENSG00000163558
COSMIC: PRKCI
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000163558 | ENST00000295797 | P41743 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA128394595 | PLCH1 | Gene | Pathway | associated | |||
| PA134875646 | PLCZ1 | Gene | Pathway | associated | |||
| PA134914471 | PLCH2 | Gene | Pathway | associated | |||
| PA33384 | PLCB1 | Gene | Pathway | associated | |||
| PA33385 | PLCB2 | Gene | Pathway | associated | |||
| PA33386 | PLCB3 | Gene | Pathway | associated | |||
| PA33387 | PLCB4 | Gene | Pathway | associated | |||
| PA33388 | PLCD1 | Gene | Pathway | associated | |||
| PA33389 | PLCD3 | Gene | Pathway | associated | |||
| PA33390 | PLCD4 | Gene | Pathway | associated | |||
| PA33391 | PLCE1 | Gene | Pathway | associated | |||
| PA33392 | PLCG1 | Gene | Pathway | associated | 20124951 | ||
| PA33393 | PLCG2 | Gene | Pathway | associated | 20124951 | ||
| PA33394 | PLCL1 | Gene | Pathway | associated | |||
| PA33395 | PLCL2 | Gene | Pathway | associated | |||
| PA34183 | RAF1 | Gene | Pathway | associated | 20124951 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 36833174 | 2023 | Investigation of UTR Variants by Computational Approaches Reveal Their Functional Significance in PRKCI Gene Regulation. | 1 |
| 37016317 | 2023 | Paired protein kinases PRKCI-RIPK2 promote pancreatic cancer growth and metastasis via enhancing NF-κB/JNK/ERK phosphorylation. | 0 |
| 38092754 | 2023 | Enhanced SREBP2-driven cholesterol biosynthesis by PKCλ/ι deficiency in intestinal epithelial cells promotes aggressive serrated tumorigenesis. | 0 |
| 36833174 | 2023 | Investigation of UTR Variants by Computational Approaches Reveal Their Functional Significance in PRKCI Gene Regulation. | 1 |
| 37016317 | 2023 | Paired protein kinases PRKCI-RIPK2 promote pancreatic cancer growth and metastasis via enhancing NF-κB/JNK/ERK phosphorylation. | 0 |
| 38092754 | 2023 | Enhanced SREBP2-driven cholesterol biosynthesis by PKCλ/ι deficiency in intestinal epithelial cells promotes aggressive serrated tumorigenesis. | 0 |
| 35124330 | 2022 | Human placenta and trophoblasts simultaneously express three isoforms of atypical protein kinase-c. | 3 |
| 35785767 | 2022 | PKCι Is a Promising Prognosis Biomarker and Therapeutic Target for Pancreatic Cancer. | 3 |
| 35124330 | 2022 | Human placenta and trophoblasts simultaneously express three isoforms of atypical protein kinase-c. | 3 |
| 35785767 | 2022 | PKCι Is a Promising Prognosis Biomarker and Therapeutic Target for Pancreatic Cancer. | 3 |
| 33146712 | 2021 | Transcription factor Sp1 is upregulated by PKCι to drive the expression of YAP1 during pancreatic carcinogenesis. | 5 |
| 33147518 | 2021 | The interruption of atypical PKC signaling and Temozolomide combination therapy against glioblastoma. | 7 |
| 33660800 | 2021 | CircPRKCI regulates proliferation, migration and cycle of lung adenocarcinoma cells by targeting miR-219a-5p-regulated CAMK1D. | 6 |
| 33880589 | 2021 | circ-PRKCI targets miR-1294 and miR-186-5p by downregulating FOXK1 expression to suppress glycolysis in hepatocellular carcinoma. | 15 |
| 34560002 | 2021 | PKCλ/ι inhibition activates an ULK2-mediated interferon response to repress tumorigenesis. | 10 |
Citation
Verline Justilien ; Alan P Fields
PRKCI (protein kinase C, iota)
Atlas Genet Cytogenet Oncol Haematol. 2012-06-01
Online version: http://atlasgeneticsoncology.org/gene/41857/prkci
