MECOM (Ecotropic Viral Integration Site 1 (EVI1) and Myelodysplastic Syndrome 1 (MDS1-EVI1)
2015-05-01 Rotraud Wieser AffiliationIdentity


Abstract
The MECOM locus in chromosome band 3q26.2 gives rise to two major mRNA and protein species, EVI1 and MDS1\/EVI1, of which the former has been characterized far more extensively. EVI1 has been implicated in the maintenance and expansion of normal hematopoietic stem cells. In addition, it plays a role as an oncogene in myeloid leukemia and in certain solid tumors, its overexpression being associated with a dismal prognosis in several of these malignancies. EVI1 exerts its biological effects mainly by regulating gene transcription, and does so by acting as a sequence specific transcription factor, by modulating the activity of other sequence specific transcription factors, and by regulating promoter CpG island methylation.
DNA/RNA
Description
The human MDS1 gene consists of 4 exons spread over a genomic region of more than 500 kb. MDS1 exon 4 is located less than 2 kb upstream of EVI1 exon1a. MDS1 can also be expressed on its own. The MDS1/EVI1 mRNA presumably results from splicing of the second exon of MDS1 to the second exon of EVI1 (Fig. 1B).
Transcription
Proteins
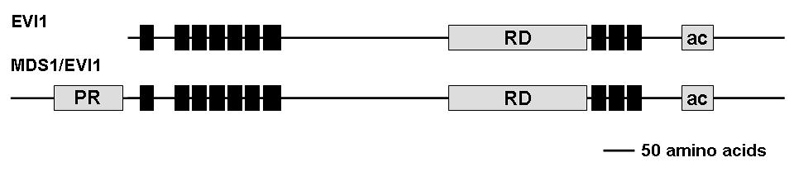
Description
Predicted translation of MDS1-EVI1 adds 188 amino acids to the N-terminus of EVI1. 63 of these additional amino acids are encoded by exon 2 and beginning of exon 3 of EVI1, and the remaining 125 from the MDS1 gene. MDS1-EVI1 contains a PR domain, which is about 40% homologous to the N-terminus of the retinoblastoma-binding protein, RIZ, and the PRDI-BF1 transcription factor. Some biological functions of MDS1/EVI1 were reported to be different from, or even antagonistic to, those of EVI1, while in other cases, EVI1 and MDS1/EVI1 acted in a similar manner. MDS1/EVI1 (PRDM3) has H3K9me1 methyltransferase activity and a role in maintaining heterochromatin integrity (Pinheiro et al, 2012).
Expression
In the adult mouse, the Evi1 mRNA is expressed, at varying levels, in the kidney, lung, stomach, ovary, uterus, intestine, thymus, spleen, heart, brain, and liver. In the mouse embryo, Evi1 mRNA levels are high in the urinary system and Mullerian ducts, the lung, the heart, and the emerging limb buds.
Similar Evi1 expression patterns were also observed in Xenopus, chicken, and zebrafish.
In human and murine hematopoiesis, EVI1 mRNA levels are high in the most immature cell populations and decline in the course of differentiation (Kataoka et al, 2011; Bindels et al, 2012; Steinleitner et al, 2012).
EVI1 expression is regulated by RUNX1 and ELK1, by retinoic acid via RAR/RXR, and by certain MLL fusion proteins (Bingemann et al, 2009; Arai et al, 2011; Maicas et al, 2013).
Localisation
Function
In the mouse, homozygous disruption of the 6th exon of the Evi1 gene led to embryonic lethality, with widespread hypocellularity, reduced body size, small or absent limb buds, a pale yolk sac and placenta, abnormal development of the nervous system and the heart, and massive haemorrhaging. (Hoyt et al, 1997). Functions of Evi1 and/or Mds1/Evi1 in heart development, spine formation, and, particularly, maintenance and expansion of hematopoietic stem cells (HSCs) have been deduced from, or confirmed through, the phenotypes of additional MECOM knockout models (Goyama et al, 2008; Kataoka et al, 2011; Zhang et al, 2011; Bard-Chapeau et al, 2014; Juneja et al, 2014). A role of Evi1 in HSCs was also corroborated through overexpression and gene marking experiments (Buonamici et al, 2004; Laricchia-Robbio et al, 2008; Dickstein et al, 2010; Kataoka et al, 2011).
Support for a role of Evi1 as a leukemia initiating and promoting oncogene has been obtained through mouse bone marrow transduction/transplantation models, in which overexpression of Evi1 alone caused a myelodysplastic syndrome (MDS) like disease, while its co-expression with other oncogenes led to AML (Buonamici et al, 2004; Jin et al, 2007; Watanabe-Okochi et al, 2008; Watanabe-Okochi et al, 2013). Even more compellingly, in a human gene therapy trial for chronic granulomatous disease, activating integrations of the therapeutic vector into the MECOM locus led to clonal expansion with progression to MDS and, ultimately, AML (Stein et al, 2010). Evi1 was proposed to be essential for AML leukemia stem cell (LSC) function since its experimental down-regulation reduced leukemogenicity in several mouse models of AML (Goyama et al, 2008; Bindels et al, 2012). Evi1 expression was also associated with leukemia initiating capacity in chronic myeloid leukemia (CML) (Sato et al, 2014). A prominent role of EVI1 in therapy resistance was suggested by a number of clinical trials, and illustrated by in vitro data demonstrating that its ectopic expression reduced, and its knockdown enhanced, cellular responsiveness to chemotherapeutic drugs (Bindels et al, 2012; Konantz et al, 2012; Yamakawa et al, 2012; Rommer et al, 2013).
Beyond its roles in normal and malignant hematopoiesis, EVI1 negatively regulated NF-kB dependent inflammation (Xu et al, 2012) and promoted adipocyte differentiation (Ishibashi et al, 2012).
EVI1 exerts its biological functions mainly by acting as a transcription factor. and regulates the expression of both protein coding and miRNA genes. Reported direct EVI1 target genes are MS4A3 (Heller et al, 2015), PLZF (Takahashi and Licht, 2002), Gata2 (Yuasa et al; 2005), Pbx1 (Shimabe et al, 2009), Pten (Yoshimi et al, 2011), Gpr56 (Saito et al, 2013), DeltaNp63 (Nayak et al, 2013), Bcl-xL (Pradhan et al, 2011), Calreticulin (Qiu et al, 2008), Ppargamma2 (Ishibashi et al, 2012), miR-1-2 (Gomez-Benito et al, 2010), miR-9 (Senyuk et al, 2013), miR-124 (Dickstein et al, 2010), and miR-449A (De Weer et al, 2011). ChIP-seq, combined with genome wide gene expression profiling, has been employed for large-scale identification of EVI1 target genes in ovarian cancer and murine myeloid cell lines (Bard-Chapeau et al, 2012; Glass et al, 2013). EVI1 associates with a number of transcriptional cofactors like HDAC1 (Vinatzer et al, 2001), CtBP1 (Palmer et al, 2001), CtBP2 (Turner and Crossley, 1998), CBP, P/CAF (Chakraborty et al, 2001), the histone methyl transferases SUV39H1 and G9a (Spensberger et al, 2008a; Goyama et al, 2010), the ATP dependent helicases BRG1 and BRM (Chi et al, 2003), and the member of histone deacetylase complex, Mbd3b (Spensberger et al, 2008b). EVI1 was also shown to interact with DNA methyl transferases (Lugthart et al, 2011; Senyuk et al, 2011), leading to methylation of CpG islands of some of its target genes, among them, CADM1 (Fisser et al, 2014), miR-9 (Senyuk et al, 2013), and miR-124 (Dickstein et al, 2010). Furthermore, EVI1 interacted with, and modulated the function of, other sequence specific transcription factors, e.g. GATA1 (Laricchia-Robbio et al, 2006), RUNX1/AML1 (Senyuk et al, 2007), PU.1 (Laricchia-Robbio et al, 2009), SMAD3 (Kurokawa et al, 1998; Izutsu et al, 2001; Alliston et al, 2005), FOS (Bard-Chapeau et al, 2012), NFkB (Xu et al, 2012), and RAR/RXR (Bingemann et al, 2009; Steinmetz et al, 2014).
In addition to its activity as a regulator of transcription, EVI1 has been reported to inhibit c-jun N-terminal kinase (Kurokawa et al, 2000), and to stimulate PI3K/AKT signalling (Liu et al, 2006; Yoshimi et al, 2011).
Homology
Implicated in
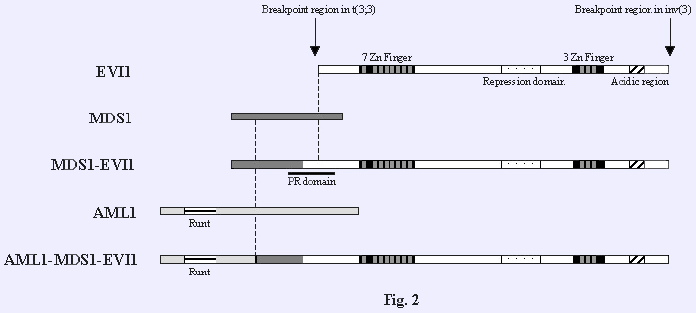
t(2;3)(p16;q26) BCL11A/MECOM in acute myeloid leukemia
t(3;21)(q26;q21) NRIP1/MECOM in acute myeloid leukemia (Haferlach et al., 2012) t(3;3)(q21;q26) LINC01565/MECOM in acute myeloid leukemia (Pekarsky et al., 1997)
t(3;7)(q26;q21) MECOM/CDK6 in myeloid leukemias
t(3;7)(q26;q34) TRB/MECOM in acute myeloid leukemia (M0-AML) (Suzukawa et al., 1999)
t(3;15)(q26;p13) MECOM/FRMD5 in lung adenocarcinoma (Yoshihara et al., 2014)
Breakpoints

Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 15849193 | 2005 | Repression of bone morphogenetic protein and activin-inducible transcription by Evi-1. | Alliston T et al |
| 17014970 | 2006 | Conservation and expression of a novel alternatively spliced Evi1 exon. | Alzuherri H et al |
| 21190993 | 2011 | Evi-1 is a transcriptional target of mixed-lineage leukemia oncoproteins in hematopoietic stem cells. | Arai S et al |
| 16014322 | 2005 | Regulation of the expression of the oncogene EVI1 through the use of alternative mRNA 5'-ends. | Aytekin M et al |
| 24586749 | 2014 | Mice carrying a hypomorphic Evi1 allele are embryonic viable but exhibit severe congenital heart defects. | Bard-Chapeau EA et al |
| 12393383 | 2003 | High EVI1 expression predicts poor survival in acute myeloid leukemia: a study of 319 de novo AML patients. | Barjesteh van Waalwijk van Doorn-Khosrovani S et al |
| 8108138 | 1994 | Induction of two alternatively spliced evi-1 proto-oncogene transcripts by cAMP in kidney cells. | Bartholomew C et al |
| 22553314 | 2012 | EVI1 is critical for the pathogenesis of a subset of MLL-AF9-rearranged AMLs. | Bindels EM et al |
| 19843176 | 2009 | Zinc finger transcription factor ecotropic viral integration site 1 is induced by all-trans retinoic acid (ATRA) and acts as a dual modulator of the ATRA response. | Bingemann SC et al |
| 17341266 | 2007 | An interphase fluorescence in situ hybridisation assay for the detection of 3q26.2/EVI1 rearrangements in myeloid malignancies. | Bobadilla D et al |
| 2113669 | 1990 | Alternative splicing of the Evi-1 zinc finger gene generates mRNAs which differ by the number of zinc finger motifs. | Bordereaux D et al |
| 15343390 | 2004 | EVI1 induces myelodysplastic syndrome in mice. | Buonamici S et al |
| 11568182 | 2001 | Interaction of EVI1 with cAMP-responsive element-binding protein-binding protein (CBP) and p300/CBP-associated factor (P/CAF) results in reversible acetylation of EVI1 and in co-localization in nuclear speckles. | Chakraborty S et al |
| 14555651 | 2003 | EVI1 promotes cell proliferation by interacting with BRG1 and blocking the repression of BRG1 on E2F1 activity. | Chi Y et al |
| 21569010 | 2011 | EVI1-mediated down regulation of MIR449A is essential for the survival of EVI1 positive leukaemic cells. | De Weer A et al |
| 22161860 | 2013 | Overexpression of Evi-1 oncoprotein represses TGF-β signaling in colorectal cancer. | Deng X et al |
| 20448201 | 2010 | Methylation and silencing of miRNA-124 by EVI1 and self-renewal exhaustion of hematopoietic stem cells in murine myelodysplastic syndrome. | Dickstein J et al |
| 16109773 | 2005 | Insertional mutagenesis identifies genes that promote the immortalization of primary bone marrow progenitor cells. | Du Y et al |
| 25491945 | 2015 | Induction of the proapoptotic tumor suppressor gene Cell Adhesion Molecule 1 by chemotherapeutic agents is repressed in therapy resistant acute myeloid leukemia. | Fisser MC et al |
| 20842122 | 2010 | EVI1 controls proliferation in acute myeloid leukaemia through modulation of miR-1-2. | Gómez-Benito M et al |
| 23826213 | 2013 | Global Identification of EVI1 Target Genes in Acute Myeloid Leukemia. | Glass C et al |
| 19776757 | 2010 | EVI-1 interacts with histone methyltransferases SUV39H1 and G9a for transcriptional repression and bone marrow immortalization. | Goyama S et al |
| 18682242 | 2008 | Evi-1 is a critical regulator for hematopoietic stem cells and transformed leukemic cells. | Goyama S et al |
| 24703711 | 2014 | A single oncogenic enhancer rearrangement causes concomitant EVI1 and GATA2 deregulation in leukemia. | Gröschel S et al |
| 18181178 | 2008 | Expression and prognostic significance of different mRNA 5'-end variants of the oncogene EVI1 in 266 patients with de novo AML: EVI1 and MDS1/EVI1 overexpression both predict short remission duration. | Haas K et al |
| 22887804 | 2012 | Three novel cytogenetically cryptic EVI1 rearrangements associated with increased EVI1 expression and poor prognosis identified in 27 acute myeloid leukemia cases. | Haferlach C et al |
| 25886616 | 2015 | EVI1 promotes tumor growth via transcriptional repression of MS4A3. | Heller G et al |
| 9256345 | 1997 | The Evi1 proto-oncogene is required at midgestation for neural, heart, and paraxial mesenchyme development. | Hoyt PR et al |
| 22473998 | 2012 | An Evi1-C/EBPβ complex controls peroxisome proliferator-activated receptor γ2 gene expression to initiate white fat cell differentiation. | Ishibashi J et al |
| 10867128 | 2000 | The EVI-1 gene--its role in pathogenesis of human leukemias. | Jółkowska J et al |
| 17227832 | 2007 | Trib1 and Evi1 cooperate with Hoxa and Meis1 in myeloid leukemogenesis. | Jin G et al |
| 24316420 | 2014 | Deletion of Mecom in mouse results in early-onset spinal deformity and osteopenia. | Juneja SC et al |
| 22084405 | 2011 | Evi1 is essential for hematopoietic stem cell self-renewal, and its expression marks hematopoietic cells with long-term multilineage repopulating activity. | Kataoka K et al |
| 16156860 | 2005 | The Evi1 proto-oncoprotein blocks endomitosis in megakaryocytes by inhibiting sustained cyclin-dependent kinase 2 catalytic activity. | Kilbey A et al |
| 22828445 | 2013 | EVI-1 modulates leukemogenic potential and apoptosis sensitivity in human acute lymphoblastic leukemia. | Konantz M et al |
| 19605700 | 2009 | Inducible expression of EVI1 in human myeloid cells causes phenotypes consistent with its role in myelodysplastic syndromes. | Konrad TA et al |
| 21493867 | 2011 | The transcription factor evi-1 is overexpressed, promotes proliferation, and is prognostically unfavorable in infratentorial ependymomas. | Koos B et al |
| 10856240 | 2000 | The evi-1 oncoprotein inhibits c-Jun N-terminal kinase and prevents stress-induced cell death. | Kurokawa M et al |
| 19208846 | 2009 | EVI1 Impairs myelopoiesis by deregulation of PU.1 function. | Laricchia-Robbio L et al |
| 8616064 | 1996 | t(2;3)(p23;q26) in a patient with AML M2. | Levaltier X et al |
| 16462766 | 2006 | Evi1 is a survival factor which conveys resistance to both TGFbeta- and taxol-mediated cell death via PI3K/AKT. | Liu Y et al |
| 20855866 | 2011 | Aberrant DNA hypermethylation signature in acute myeloid leukemia directed by EVI1. | Lugthart S et al |
| 22689058 | 2013 | Functional characterization of the promoter region of the human EVI1 gene in acute myeloid leukemia: RUNX1 and ELK1 directly regulate its transcription. | Maicas M et al |
| 15905132 | 2005 | Evi-1 expression in Xenopus. | Mead PE et al |
| 21636719 | 2011 | Fanconi anemia (FA)-associated 3q gains in leukemic transformation consistently target EVI1, but do not affect low TERC expression in FA. | Meyer S et al |
| 17243162 | 2007 | Amplification and translocation of 3q26 with overexpression of EVI1 in Fanconi anemia-derived childhood acute myeloid leukemia with biallelic FANCD1/BRCA2 disruption. | Meyer S et al |
| 8313895 | 1994 | Generation of the AML1-EVI-1 fusion gene in the t(3;21)(q26;q22) causes blastic crisis in chronic myelocytic leukemia. | Mitani K et al |
| 2115646 | 1990 | Unique expression of the human Evi-1 gene in an endometrial carcinoma cell line: sequence of cDNAs and structure of alternatively spliced transcripts. | Morishita K et al |
| 17409414 | 2007 | Amplification of MDS1/EVI1 and EVI1, located in the 3q26.2 amplicon, is associated with favorable patient prognosis in ovarian cancer. | Nanjundan M et al |
| 23665236 | 2013 | EVI1 targets ΔNp63 and upregulates the cyclin dependent kinase inhibitor p21 independent of p53 to delay cell cycle progression and cell proliferation in colon cancer cells. | Nayak KB et al |
| 9447815 | 1997 | The EVI1 gene in myeloid leukemia. | Nucifora G et al |
| 8171026 | 1994 | Consistent intergenic splicing and production of multiple transcripts between AML1 at 21q22 and unrelated genes at 3q26 in (3;21)(q26;q22) translocations. | Nucifora G et al |
| 16582916 | 2006 | Correction of X-linked chronic granulomatous disease by gene therapy, augmented by insertional activation of MDS1-EVI1, PRDM16 or SETBP1. | Ott MG et al |
| 11328817 | 2001 | Evi-1 transforming and repressor activities are mediated by CtBP co-repressor proteins. | Palmer S et al |
| 21057539 | 2011 | Control of EVI-1 oncogene expression in metastatic breast cancer cells through microRNA miR-22. | Patel JB et al |
| 9044825 | 1997 | Fusion of ETV6 to MDS1/EVI1 as a result of t(3;12)(q26;p13) in myeloproliferative disorders. | Peeters P et al |
| 9307271 | 1997 | Activation of a novel gene in 3q21 and identification of intergenic fusion transcripts with ecotropic viral insertion site I in leukemia. | Pekarsky Y et al |
| 1893871 | 1991 | Patterns of Evi-1 expression in embryonic and adult tissues suggest that Evi-1 plays an important regulatory role in mouse development. | Perkins AS et al |
| 22939622 | 2012 | Prdm3 and Prdm16 are H3K9me1 methyltransferases required for mammalian heterochromatin integrity. | Pinheiro I et al |
| 16342172 | 2006 | EVI1 is consistently expressed as principal transcript in common and rare recurrent 3q26 rearrangements. | Poppe B et al |
| 21980434 | 2011 | Acetylation of the proto-oncogene EVI1 abrogates Bcl-xL promoter binding and induces apoptosis. | Pradhan AK et al |
| 18303859 | 2008 | Regulation of the calreticulin gene by GATA6 and Evi-1 transcription factors. | Qiu Y et al |
| 8695816 | 1996 | Fluorescence in situ hybridization analysis of t(3; 12)(q26; p13): a recurring chromosomal abnormality involving the TEL gene (ETV6) in myelodysplastic syndromes. | Raynaud SD et al |
| 23457546 | 2013 | EVI1 inhibits apoptosis induced by antileukemic drugs via upregulation of CDKN1A/p21/WAF in human myeloid cells. | Rommer A et al |
| 8049440 | 1994 | Expression of EVI1 in myelodysplastic syndromes and other hematologic malignancies without 3q26 translocations. | Russell M et al |
| 23478665 | 2013 | Maintenance of the hematopoietic stem cell pool in bone marrow niches by EVI1-regulated GPR56. | Saito Y et al |
| 24747972 | 2014 | Evi1 defines leukemia-initiating capacity and tyrosine kinase inhibitor resistance in chronic myeloid leukemia. | Sato T et al |
| 21695170 | 2011 | The oncoprotein EVI1 and the DNA methyltransferase Dnmt3 co-operate in binding and de novo methylation of target DNA. | Senyuk V et al |
| 17575132 | 2007 | Repression of RUNX1 activity by EVI1: a new role of EVI1 in leukemogenesis. | Senyuk V et al |
| 23509296 | 2013 | Critical role of miR-9 in myelopoiesis and EVI1-induced leukemogenesis. | Senyuk V et al |
| 19767769 | 2009 | Pbx1 is a downstream target of Evi-1 in hematopoietic stem/progenitors and leukemic cells. | Shimabe M et al |
| 20363750 | 2010 | Acetylation of lysine 564 adjacent to the C-terminal binding protein-binding motif in EVI1 is crucial for transcriptional activation of GATA2. | Shimahara A et al |
| 23770046 | 2013 | SUMO1 negatively regulates the transcriptional activity of EVI1 and significantly increases its co-localization with EVI1 after treatment with arsenic trioxide. | Singh S et al |
| 18619962 | 2008 | A novel interaction between the proto-oncogene Evi1 and histone methyltransferases, SUV39H1 and G9a. | Spensberger D et al |
| 20098431 | 2010 | Genomic instability and myelodysplasia with monosomy 7 consequent to EVI1 activation after gene therapy for chronic granulomatous disease. | Stein S et al |
| 23155256 | 2012 | EVI1 and MDS1/EVI1 expression during primary human hematopoietic progenitor cell differentiation into various myeloid lineages. | Steinleitner K et al |
| 25486480 | 2014 | The oncogene EVI1 enhances transcriptional and biological responses of human myeloid cells to all-trans retinoic acid. | Steinmetz B et al |
| 15085164 | 2004 | Translocation t(2;3)(p15-23;q26-27) in myeloid malignancies: report of 21 new cases, clinical, cytogenetic and molecular genetic features. | Stevens-Kroef M et al |
| 24828867 | 2015 | Aberrant expression of ecotropic viral integration site-1 in acute myeloid leukemia and acute lymphoblastic leukemia. | Su G et al |
| 10482986 | 1999 | Activation of EVI1 transcripts with chromosomal translocation joining the TCRVbeta locus and the EVI1 gene in human acute undifferentiated leukemia cell line (Kasumi-3) with a complex translocation of der(3)t(3;7;8). | Suzukawa K et al |
| 12200691 | 2002 | The human promyelocytic leukemia zinc finger gene is regulated by the Evi-1 oncoprotein and a novel guanine-rich site binding protein. | Takahashi S et al |
| 23752186 | 2014 | EVI1 oncogene promotes KRAS pathway through suppression of microRNA-96 in pancreatic carcinogenesis. | Tanaka M et al |
| 9724649 | 1998 | Cloning and characterization of mCtBP2, a co-repressor that associates with basic Krüppel-like factor and other mammalian transcriptional regulators. | Turner J et al |
| 16574097 | 2006 | Evi1 is specifically expressed in the distal tubule and duct of the Xenopus pronephros and plays a role in its formation. | Van Campenhout C et al |
| 25936528 | 2015 | The regulatory interaction of EVI1 with the TCL1A oncogene impacts cell survival and clinical outcome in CLL. | Vasyutina E et al |
| 12461752 | 2003 | Quantitative comparison of the expression of EVI1 and its presumptive antagonist, MDS1/EVI1, in patients with myeloid leukemia. | Vinatzer U et al |
| 12555218 | 2003 | Acute myelogenous leukemia with the t(3;12)(q26;p13) translocation: case report and review of the literature. | Voutsadakis IA et al |
| 23547050 | 2013 | The shortest isoform of C/EBPβ, liver inhibitory protein (LIP), collaborates with Evi1 to induce AML in a mouse BMT model. | Watanabe-Okochi N et al |
| 23776681 | 2013 | Phosphorylation of the leukemic oncoprotein EVI1 on serine 196 modulates DNA binding, transcriptional repression and transforming ability. | White DJ et al |
| 17507183 | 2007 | The oncogene and developmental regulator EVI1: expression, biochemical properties, and biological functions. | Wieser R et al |
| 12551823 | 2003 | Interphase fluorescence in situ hybridization assay for the detection of rearrangements of the EVI-1 locus in chromosome band 3q26 in myeloid malignancies. | Wieser R et al |
| 22581859 | 2012 | EVI1 acts as an inducible negative-feedback regulator of NF-κB by inhibiting p65 acetylation. | Xu X et al |
| 22295105 | 2012 | The increased expression of integrin α6 (ITGA6) enhances drug resistance in EVI1(high) leukemia. | Yamakawa N et al |
| 24703906 | 2014 | A remote GATA2 hematopoietic enhancer drives leukemogenesis in inv(3)(q21;q26) by activating EVI1 expression. | Yamazaki H et al |
| 16532439 | 2006 | t(3;21)(q26;q22) in myeloid leukemia: an aggressive syndrome of blast transformation associated with hydroxyurea or antimetabolite therapy. | Yin CC et al |
| 25500544 | 2015 | The landscape and therapeutic relevance of cancer-associated transcript fusions. | Yoshihara K et al |
| 21289308 | 2011 | Evi1 represses PTEN expression and activates PI3K/AKT/mTOR via interactions with polycomb proteins. | Yoshimi A et al |
| 15889140 | 2005 | Oncogenic transcription factor Evi1 regulates hematopoietic stem cell proliferation through GATA-2 expression. | Yuasa H et al |
| 8813093 | 1996 | Expression of EVI1 and the Retinoblastoma genes in acute myelogenous leukemia with t(3;13)(q26;q13-14). | Yufu Y et al |
| 21666053 | 2011 | PR-domain-containing Mds1-Evi1 is critical for long-term hematopoietic stem cell function. | Zhang Y et al |
Other Information
Locus ID:
NCBI: 2122
MIM: 165215
HGNC: 3498
Ensembl: ENSG00000085276
Variants:
dbSNP: 2122
ClinVar: 2122
TCGA: ENSG00000085276
COSMIC: MECOM
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 36269819 | 2023 | EVI1 exerts distinct roles in AML via ERG and cyclin D1 promoting a chemoresistant and immune-suppressive environment. | 1 |
| 36282419 | 2023 | EVI1 upregulates PTGS1 (COX1) and decreases the action of tyrosine kinase inhibitors (TKIs) in chronic myeloid leukemia cells. | 1 |
| 36358022 | 2023 | The adverse impact of ecotropic viral integration site-1 (EVI1) overexpression on the prognosis of acute myeloid leukemia with KMT2A gene rearrangement in different risk stratification subtypes. | 1 |
| 37067177 | 2023 | Expanded phenotypic and hematologic abnormalities beyond bone marrow failure in MECOM-associated syndromes. | 3 |
| 37185814 | 2023 | Epigenetic landscape reveals MECOM as an endothelial lineage regulator. | 6 |
| 37230770 | 2023 | Perinatal-lethal nonimmune fetal hydrops attributed to MECOM-associated bone marrow failure. | 0 |
| 37407873 | 2023 | MECOM Deficiency: from Bone Marrow Failure to Impaired B-Cell Development. | 3 |
| 37610030 | 2023 | A novel mutation in MECOM affects MPL regulation in vitro and results in thrombocytopenia and bone marrow failure. | 1 |
| 36269819 | 2023 | EVI1 exerts distinct roles in AML via ERG and cyclin D1 promoting a chemoresistant and immune-suppressive environment. | 1 |
| 36282419 | 2023 | EVI1 upregulates PTGS1 (COX1) and decreases the action of tyrosine kinase inhibitors (TKIs) in chronic myeloid leukemia cells. | 1 |
| 36358022 | 2023 | The adverse impact of ecotropic viral integration site-1 (EVI1) overexpression on the prognosis of acute myeloid leukemia with KMT2A gene rearrangement in different risk stratification subtypes. | 1 |
| 37067177 | 2023 | Expanded phenotypic and hematologic abnormalities beyond bone marrow failure in MECOM-associated syndromes. | 3 |
| 37185814 | 2023 | Epigenetic landscape reveals MECOM as an endothelial lineage regulator. | 6 |
| 37230770 | 2023 | Perinatal-lethal nonimmune fetal hydrops attributed to MECOM-associated bone marrow failure. | 0 |
| 37407873 | 2023 | MECOM Deficiency: from Bone Marrow Failure to Impaired B-Cell Development. | 3 |
Citation
Rotraud Wieser
MECOM (Ecotropic Viral Integration Site 1 (EVI1) and Myelodysplastic Syndrome 1 (MDS1-EVI1)
Atlas Genet Cytogenet Oncol Haematol. 2015-05-01
Online version: http://atlasgeneticsoncology.org/gene/19/mecom
Historical Card
2007-12-01 MECOM (Ecotropic Viral Integration Site 1 (EVI1) and Myelodysplastic Syndrome 1 (MDS1-EVI1) by Rotraud Wieser Affiliation
2003-05-01 MECOM (Ecotropic Viral Integration Site 1 (EVI1) and Myelodysplastic Syndrome 1 (MDS1-EVI1) by Soumen Chakraborty,Silvia Buonamici,Vitalyi Senyuk,Giuseppina Nucifora Affiliation
