T-lineage acute lymphoblastic leukemia (T-ALL)
2007-05-01 Susana C Raimondi Affiliation1.Department of pathology, St. Jude Childrens Research Hospital, 332 North Lauderdale Street, Memphis, Tennessee 38105-2794, USA
Clinics and Pathology
Etiology
The TCR promoter and enhancer elements are juxtaposed to a relatively small number of developmentally important genes that encode transcription factors leading to T-cell malignancies. The chromosomal aberrations that affect the TCR loci were among the first to be reported in T-ALL. Subsequently, these and other rarer translocations facilitated the identification of genes that are altered in T-ALL, many of which are also transcriptionally activated without evidence of any detectable chromosomal rearrangement affecting these loci. In summary, the ectopic expression of TAL1(SCL),LYL1, LMO1, LMO2, TLX1(HOX11), and TLX3 (HOX11L2), NOTCH1-activating mutations, and CDKN2-inactivating deletions are among the most prevalent causes of human T-ALL.
Epidemiology
A high number of T-ALL cases have cryptic abnormalities, as shown by fluorescent in situ hybridization (FISH) or other molecular methods. In some instances, this occurs because some of the loci involved in oncogenic rearrangements of T-ALL have a near-telomeric location that generates subtle exchanges in DNA material, and these changes subsequently cause the cryptic translocations. As many as 80% of patients with T-ALL have cryptic deletions of the putative tumor suppressor gene CDKN2A (INK4A) (9p21), and as many as 60% have cryptic deletions of TAL1 (1p32). Other genes such as TLX1 (HOX11) (10q24) and NOTCH1 (9q34) are activated at a much higher frequency than expected from cytogenetic studies alone; thus, simultaneous dysregulation of different signaling pathways may contribute to the multistep pathogenesis of T-ALL subgroups.
Clinics
Prognosis
At present, there are no genetic markers in T-ALL that reliably predict treatment response or outcome. Gene expression analysis has revealed the prognostic significance of T-ALL oncogenes and the stage of thymocyte differentiation in which they are expressed. Some genetic markers have been shown to be of clinical relevance in a small series of pediatric patients with T-ALL: TLX1(HOX11)+ was associated with favorable outcome, and TAL1+ and LYL1+ were associated with unfavorable outcome. A favorable prognosis was also found with TLX1(HOX11)+ in adult T-ALL, possibly due to downregulation of antiapoptotic genes. The poor prognosis associated with T-ALL subtypes expressing TAL1 or LYL1 is thought to be caused by the concomitant upregulation of antiapoptotic genes that confer resistance to chemotherapy.
In early studies, the overexpression of TLX3(HOX11L2) was associated with poor prognosis; however, similar, more recent studies have not confirmed such findings. This difference is probably a reflection of the current aggressive treatments that have improved the therapeutic response in this subgroup of T-ALL. Therefore, the clinical significance of genetic lesions in T-ALL remains largely unknown. The prognostic significance of T-ALL subtypes most likely depends on the type and intensity of the treatment administered. The development of targeted therapy for T-ALL might be contentious, given the simultaneous presence and the high prevalence of some genetic lesions affecting T-ALL.
Cytogenetics
Note
Cytogenetics morphological
TRB@ (7q34)
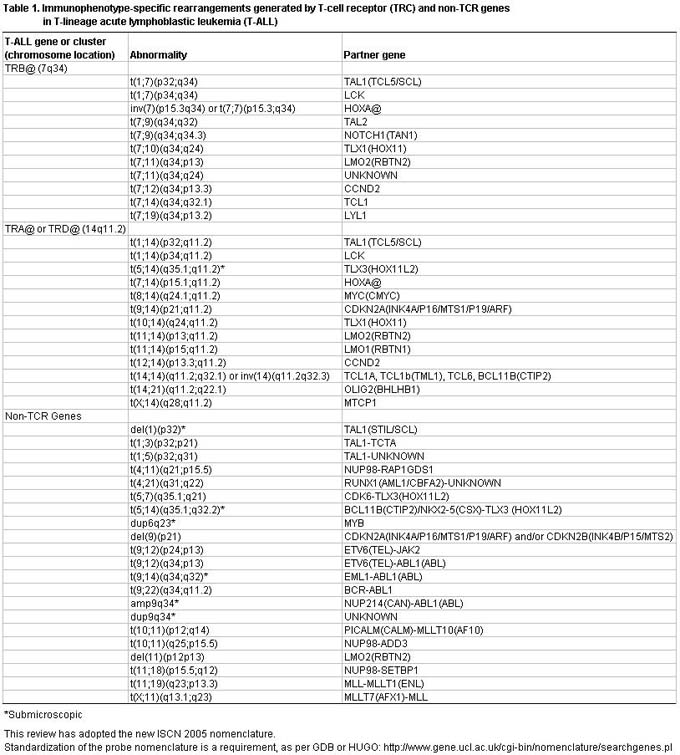
Conventional cytogenetic analysis revealed that chromosomal abnormalities affecting 7q34 (TRB@) occur in 5% to 8% of T-ALL cases with an abnormal karyotype. Recent molecular cytogenetics studies have revealed a higher incidence of TRB@ locus rearrangements (about 20% of all T-ALL cases). This finding demonstrates that the frequency of TRB@ rearrangements is similar to that of TRA@ (14q11.2) (about 24% of all T-ALL cases). Simultaneous rearrangements targeting both the TRB@ and TRA@ loci were observed in five of 126 (4%) patients with T-ALL, possibly reflecting the higher susceptibility for errors in VDJ recombination.
TRG@ (7p14))
The TRG@(TCRG) locus (7p14) may be restricted to T-cell tumors in patients with ataxia telangiectasia. The TRG@ locus is not involved in translocations in T-ALL. Historically, it was thought that the inv(7)(p15;q34) or t(7;7)(p15;q34) juxtaposed TRB@ to TRG@, but recently it was shown that the TRB@-HOXA fusion is generated by such rearrangements. One exception was a t(1;7)(p31-32;p13) in a 16 year-old male patient who had precursor T-cell lymphoblastic leukemia/lymphoma. This rearrangement was identified during the evaluation of FISH probes to detect TCR breakpoints. It was speculated that the breakpoint at 1p may have involved the TAL1 or LCK oncogenes.
TRA@ or TRD@ (14q11.2)
Among the most common chromosomal abnormalities observed by conventional cytogenetics, those associated with T-ALL are chromosome 14 alterations in which the breakpoint is located at 14q11.2. By conventional cytogenetics, this abnormality represents about 17% of all T-ALL cases (Table 1). By molecular cytogenetics studies, the incidence of TRA@/TRD@ rearrangements is about 24% of all T-ALL cases.
NON-TCR GENETIC LESIONS IN T-ALL
Summary:
In addition to conventional cytogenetic studies, molecular techniques that are more reliable, rapid, and sensitive are needed to detect multiple genetic lesions associated with T-ALL. The improved characterization of T-ALL genetic subgroups may facilitate the development of targeted gene therapy for those patients with refractory disease and less toxic therapy for those with responsive disease.
In recent years, the introduction of more intensive therapy has improved the overall outcome of patients with T-ALL, as indicated in the following landmark discoveries: Most of the recurrent abnormalities in T-ALL are different from those associated with B-lineage ALL.
Genes Involved and Proteins
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16424865 | 2006 | A t(8;9) translocation with PCM1-JAK2 fusion in a patient with T-cell lymphoma. | Adélaïde J et al |
| 2247063 | 1990 | The SCL gene is formed from a transcriptionally complex locus. | Aplan PD et al |
| 7728759 | 1995 | Cloning and characterization of TCTA, a gene located at the site of a t(1;3) translocation. | Aplan PD et al |
| 2255914 | 1990 | Disruption of the human SCL locus by "illegitimate" V-(D)-J recombinase activity. | Aplan PD et al |
| 10749961 | 2000 | Outcome of treatment in children with Philadelphia chromosome-positive acute lymphoblastic leukemia. | Aricò M et al |
| 16155013 | 2005 | Molecular genetics of acute lymphoblastic leukemia. | Armstrong SA et al |
| 12676784 | 2003 | CALM-AF10 is a common fusion transcript in T-ALL and is specific to the TCRgammadelta lineage. | Asnafi V et al |
| 15674415 | 2005 | NUP214-ABL1 amplification in t(5;14)/HOX11L2-positive ALL present with several forms and may have a prognostic significance. | Ballerini P et al |
| 15057249 | 2004 | Amplification of the ABL gene in T-cell acute lymphoblastic leukemia. | Barber KE et al |
| 17327603 | 2007 | Favorable outcome for adolescents with acute lymphoblastic leukemia treated on Dana-Farber Cancer Institute Acute Lymphoblastic Leukemia Consortium Protocols. | Barry E et al |
| 7605997 | 1995 | Does activation of the TAL1 gene occur in a majority of patients with T-cell acute lymphoblastic leukemia? A pediatric oncology group study. | Bash RO et al |
| 11672780 | 2001 | B-cell acute lymphoblastic leukemia with tandem t(14;14)(q11;q32). | Berger R et al |
| 16572206 | 2006 | HOXA cluster deregulation in T-ALL associated with both a TCRD-HOXA and a CALM-AF10 chromosomal translocation. | Bergeron J et al |
| 8412327 | 1993 | Molecular analysis of T-cell receptor transcripts in a human T-cell leukemia bearing a t(1;14) and an inv(7); cell surface expression of a TCR-beta chain in the absence of alpha chain. | Bernard O et al |
| 1886719 | 1991 | Two site-specific deletions and t(1;14) translocation restricted to human T-cell acute leukemias disrupt the 5' part of the tal-1 gene. | Bernard O et al |
| 11587205 | 2001 | A new recurrent and specific cryptic translocation, t(5;14)(q35;q32), is associated with expression of the Hox11L2 gene in T acute lymphoblastic leukemia. | Bernard OA et al |
| 16213363 | 2005 | ABL1 amplification in T-cell acute lymphoblastic leukemia. | Bernasconi P et al |
| 12661005 | 2003 | CDKN2A, CDKN2B, and MTAP gene dosage permits precise characterization of mono- and bi-allelic 9p21 deletions in childhood acute lymphoblastic leukemia. | Bertin R et al |
| 16614245 | 2006 | Activating NOTCH1 mutations predict favorable early treatment response and long-term outcome in childhood precursor T-cell lymphoblastic leukemia. | Breit S et al |
| 1913594 | 1991 | Inversions and tandem translocations involving chromosome 14q11 and 14q32 in T-prolymphocytic leukemia and T-cell leukemias in patients with ataxia telangiectasia. | Brito-Babapulle V et al |
| 16873673 | 2006 | NUP214-ABL1 in adult T-ALL: the GMALL study group experience. | Burmeister T et al |
| 17039236 | 2007 | Clinical, cytogenetic and molecular characteristics of 14 T-ALL patients carrying the TCRbeta-HOXA rearrangement: a study of the Groupe Francophone de Cytogénétique Hématologique. | Cauwelier B et al |
| 14504110 | 2004 | Clinical significance of HOX11L2 expression linked to t(5;14)(q35;q32), of HOX11 expression, and of SIL-TAL fusion in childhood T-cell malignancies: results of EORTC studies 58881 and 58951. | Cavé H et al |
| 16270038 | 2006 | Cyclin D2 dysregulation by chromosomal translocations to TCR loci in T-cell acute lymphoblastic leukemias. | Clappier E et al |
| 3194425 | 1988 | Juxtaposition of the T-cell receptor alpha-chain locus (14q11) and a region (14q32) of potential importance in leukemogenesis by a 14;14 translocation in a patient with T-cell chronic lymphocytic leukemia and ataxia-telangiectasia. | Davey MP et al |
| 17024111 | 2006 | Transition from EML1-ABL1 to NUP214-ABL1 positivity in a patient with acute T-lymphoblastic leukemia. | De Keersmaecker K et al |
| 12860018 | 2003 | t(4;11)(q21;p15), including one complex translocation t(1;4;11)(p32;q21;p15), in adult T-cell acute lymphoblastic leukemia. | Douet-Guilbert N et al |
| 9616163 | 1998 | MLL and CALM are fused to AF10 in morphologically distinct subsets of acute leukemia with translocation t(10;11): both rearrangements are associated with a poor prognosis. | Dreyling MH et al |
| 3485458 | 1986 | A new translocation, t(10;14)(q24;q11), in T cell neoplasia. | Dubé ID et al |
| 1831692 | 1991 | TAN-1, the human homolog of the Drosophila notch gene, is broken by chromosomal translocations in T lymphoblastic neoplasms. | Ellisen LW et al |
| 3486470 | 1986 | Deregulation of c-myc by translocation of the alpha-locus of the T-cell receptor in T-cell leukemias. | Erikson J et al |
| 14975618 | 2004 | Prognostic importance of TLX1 (HOX11) oncogene expression in adults with T-cell acute lymphoblastic leukaemia. | Ferrando AA et al |
| 8247530 | 1993 | The chromosomal translocation t(X;14)(q28;q11) in T-cell pro-lymphocytic leukaemia breaks within one gene and activates another. | Fisch P et al |
| 1824262 | 1991 | c-tal, a helix-loop-helix protein, is juxtaposed to the T-cell receptor-beta chain gene by a reciprocal chromosomal translocation: t(1;7)(p32;q35). | Fitzgerald TJ et al |
| 9713995 | 1998 | Deregulated expression of the TAL1 gene by t(1;5)(p32;31) in patient with T-cell acute lymphoblastic leukemia. | François S et al |
| 12682631 | 2003 | Molecular cytogenetic detection of chromosomal breakpoints in T-cell receptor gene loci. | Gesk S et al |
| 14512392 | 2003 | Childhood T-cell acute lymphoblastic leukemia: the Dana-Farber Cancer Institute acute lymphoblastic leukemia consortium experience. | Goldberg JM et al |
| 11423618 | 2001 | Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. | Gorre ME et al |
| 15990867 | 2005 | Significance of HOX11L2/TLX3 expression in children with T-cell acute lymphoblastic leukemia treated on Children's Cancer Group protocols. | Gottardo NG et al |
| 16612405 | 2006 | Notch 1 activation in the molecular pathogenesis of T-cell acute lymphoblastic leukaemia. | Grabher C et al |
| 15361874 | 2004 | Fusion of NUP214 to ABL1 on amplified episomes in T-cell acute lymphoblastic leukemia. | Graux C et al |
| 9315101 | 1997 | Alternative origin of p13MTCP1-encoding transcripts in mature T-cell proliferations with t(X;14) translocations. | Gritti C et al |
| 14564000 | 2003 | LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. | Hacein-Bey-Abina S et al |
| 12399963 | 2002 | Disruption of the RanBP17/Hox11L2 region by recombination with the TCRdelta locus in acute lymphoblastic leukemias with t(5;14)(q34;q11). | Hansen-Hagge TE et al |
| 2207332 | 1990 | Abnormalities of the long arm of chromosome 6 in childhood acute lymphoblastic leukemia. | Hayashi Y et al |
| 12066788 | 2002 | Recurrent involvement of the MLL gene in adult T-lineage acute lymphoblastic leukemia. | Hayette S et al |
| 10784391 | 2000 | Clinical significance of deletions of chromosome arm 6q in childhood acute lymphoblastic leukemia: a report from the Children's Cancer Group. | Heerema NA et al |
| 10477737 | 1999 | The (4;11)(q21;p15) translocation fuses the NUP98 and RAP1GDS1 genes and is recurrent in T-cell acute lymphocytic leukemia. | Hussey DJ et al |
| 11369658 | 2001 | Cytogenetic analysis and clinical significance in adult T-cell leukemia/lymphoma: a study of 50 cases from the human T-cell leukemia virus type-1 endemic area, Nagasaki. | Itoyama T et al |
| 12750702 | 2003 | Expression of HOX11 in childhood T-lineage acute lymphoblastic leukaemia can occur in the absence of cytogenetic aberration at 10q24: a study from the Children's Cancer Group (CCG). | Kees UR et al |
| 9360930 | 1997 | A TEL-JAK2 fusion protein with constitutive kinase activity in human leukemia. | Lacronique V et al |
| 17435759 | 2007 | Duplication of the MYB oncogene in T cell acute lymphoblastic leukemia. | Lahortiga I et al |
| 1385638 | 1992 | Pediatric leukemia/lymphoma with t(8;14)(q24;q11). | Lange BJ et al |
| 15262434 | 2004 | IGH gene involvement in two cases of acute lymphoblastic leukemia with t(14;14)(q11;q32) identified by sequential R-banding and fluorescence in situ hybridization. | Liu S et al |
| 12357357 | 2002 | Partial deletions of long arm of chromosome 6: biologic and clinical implications in adult acute lymphoblastic leukemia. | Mancini M et al |
| 16424867 | 2006 | High incidence of Notch-1 mutations in adult patients with T-cell acute lymphoblastic leukemia. | Mansour MR et al |
| 12454747 | 2002 | High incidence of Hox11L2 expression in children with T-ALL. | Mauvieux L et al |
| 14985489 | 2004 | Activation of the T-cell oncogene LMO2 after gene therapy for X-linked severe combined immunodeficiency. | McCormack MP et al |
| 2752424 | 1989 | lyl-1, a novel gene altered by chromosomal translocation in T cell leukemia, codes for a protein with a helix-loop-helix DNA binding motif. | Mellentin JD et al |
| 9558398 | 1998 | 6q deletions in acute lymphoblastic leukemia and non-Hodgkin's lymphomas. | Merup M et al |
| 12072207 | 2002 | A new translocation that rearranges the AML1 gene in a patient with T-cell acute lymphoblastic leukemia. | Mikhail FM et al |
| 12229886 | 2002 | Involvement of the MLL gene in T-lineage acute lymphoblastic leukemia. | Moorman AV et al |
| 14500364 | 2003 | The cardiac homeobox gene NKX2-5 is deregulated by juxtaposition with BCL11B in pediatric T-ALL cell lines via a novel t(5;14)(q35.1;q32.2). | Nagel S et al |
| 17308084 | 2007 | Activation of TLX3 and NKX2-5 in t(5;14)(q35;q32) T-cell acute lymphoblastic leukemia by remote 3'-BCL11B enhancers and coregulation by PU.1 and HMGA1. | Nagel S et al |
| 10778944 | 2000 | Universal inactivation of both p16 and p15 but not downstream components is an essential event in the pathogenesis of T-cell acute lymphoblastic leukemia. | Omura-Minamisawa M et al |
| 16688224 | 2006 | CUTLL1, a novel human T-cell lymphoma cell line with t(7;9) rearrangement, aberrant NOTCH1 activation and high sensitivity to gamma-secretase inhibitors. | Palomero T et al |
| 17233820 | 2007 | Fusion of NUP98 and the SET binding protein 1 (SETBP1) gene in a paediatric acute T cell lymphoblastic leukaemia with t(11;18)(p15;q12). | Panagopoulos I et al |
| 16154840 | 2005 | Novel cryptic chromosomal rearrangements in childhood acute lymphoblastic leukemia detected by multiple color fluorescent in situ hybridization. | Poppe B et al |
| 16531254 | 2006 | The effect of a novel recombination between the homeobox gene NKX2-5 and the TRD locus in T-cell acute lymphoblastic leukemia on activation of the NKX2-5 gene. | Przybylski GK et al |
| 15939259 | 2005 | Quest for effective agents to combat T-cell acute lymphoblastic leukemia. | Pui CH et al |
| 12682627 | 2003 | Clinical heterogeneity in childhood acute lymphoblastic leukemia with 11q23 rearrangements. | Pui CH et al |
| 15870488 | 2005 | Philadelphia-chromosome-positive T-lymphoblastic leukemia: acute leukemia or chronic myelogenous leukemia blastic crisis. | Raanani P et al |
| 3263151 | 1988 | Cytogenetics of childhood T-cell leukemia. | Raimondi SC et al |
| 10458233 | 1999 | Childhood acute lymphoblastic leukemia with the MLL-ENL fusion and t(11;19)(q23;p13.3) translocation. | Rubnitz JE et al |
| 10851082 | 2000 | Identification of the TCL6 genes within the breakpoint cluster region on chromosome 14q32 in T-cell leukemia. | Saitou M et al |
| 11001909 | 2000 | New recurring cytogenetic abnormalities and association of blast cell karyotypes with prognosis in childhood T-cell acute lymphoblastic leukemia: a pediatric oncology group report of 343 cases. | Schneider NR et al |
| 9408752 | 1997 | Molecular analysis of the t(8;14)(q24;q11) chromosomal breakpoint junctions in the T-cell leukemia line MOLT-16. | Shima-Rich EA et al |
| 15921375 | 2005 | Analysis of balanced rearrangements of chromosome 6 in acute leukemia: clustered breakpoints in q22-q23 and possible involvement of c-MYB in a new recurrent translocation, t(6;7)(q23;q32 through 36). | Sinclair P et al |
| 15774621 | 2005 | HOXA genes are included in genetic and biologic networks defining human acute T-cell leukemia (T-ALL). | Soulier J et al |
| 15674412 | 2005 | A new recurrent inversion, inv(7)(p15q34), leads to transcriptional activation of HOXA10 and HOXA11 in a subset of T-cell acute lymphoblastic leukemias. | Speleman F et al |
| 16015385 | 2005 | Fusion of NUP214 to ABL1 on amplified episomes in T-ALL--implications for treatment. | Stergianou K et al |
| 15334547 | 2004 | Various types of rearrangements target TLX3 locus in T-cell acute lymphoblastic leukemia. | Su XY et al |
| 16926283 | 2006 | HOX11L2/TLX3 is transcriptionally activated through T-cell regulatory elements downstream of BCL11B as a result of the t(5;14)(q35;q32). | Su XY et al |
| 10344735 | 1999 | Identification of the TCL1/MTCP1-like 1 (TML1) gene from the region next to the TCL1 locus. | Sugimoto J et al |
| 9635588 | 1998 | Frequent loss of heterozygosity on the long arm of chromosome 6: identification of two distinct regions of deletion in childhood acute lymphoblastic leukemia. | Takeuchi S et al |
| 1680958 | 1991 | Chromosomal translocations joining LCK and TCRB loci in human T cell leukemia. | Tycko B et al |
| 11170285 | 2001 | Molecular cytogenetic and clinical findings in ETV6/ABL1-positive leukemia. | Van Limbergen H et al |
| 16873670 | 2006 | The cryptic chromosomal deletion del(11)(p12p13) as a new activation mechanism of LMO2 in pediatric T-cell acute lymphoblastic leukemia. | Van Vlierberghe P et al |
| 7809072 | 1994 | Identification of the TCL1 gene involved in T-cell malignancies. | Virgilio L et al |
| 10737801 | 2000 | The t(14;21)(q11.2;q22) chromosomal translocation associated with T-cell acute lymphoblastic leukemia activates the BHLHB1 gene. | Wang J et al |
| 15472075 | 2004 | Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. | Weng AP et al |
| 16364773 | 2006 | Trisomy 4 as sole karyotypic abnormality in acute lymphoblastic leukemia: different clinical features and treatment response between B and T phenotypes? | Yip SF et al |
| 16956820 | 2006 | The outcome of molecular-cytogenetic subgroups in pediatric T-cell acute lymphoblastic leukemia: a retrospective study of patients treated according to DCOG or COALL protocols. | van Grotel M et al |
| 16673019 | 2006 | A new recurrent 9q34 duplication in pediatric T-cell acute lymphoblastic leukemia. | van Vlierberghe P et al |
| 15042105 | 2004 | Split-signal FISH for detection of chromosome aberrations in acute lymphoblastic leukemia. | van der Burg M et al |
Summary
Note
Citation
Susana C Raimondi
T-lineage acute lymphoblastic leukemia (T-ALL)
Atlas Genet Cytogenet Oncol Haematol. 2007-05-01
Online version: http://atlasgeneticsoncology.org/haematological/1374/t-lineage-acute-lymphoblastic-leukemia-(t-all)
