Therapy-Related Hematopoietic Neoplasia
2020-11-01 Mark A Micale Affiliation1.Departments of Pathology and Laboratory Medicine and Obstetrics & Gynecology Beaumont Health and Oakland University William Beaumont School of Medicine 3601 West Thirteen Mile Rd. Royal Oak, MI 48073 [email protected]
Abstract
Despite new treatments for solid tumors and hematolymphoid malignancies, including targeted therapy and immunotherapy, many cancer patients still require more traditional therapies utilizing cytotoxic chemotherapy and conventional ionizing radiation. These treatments can result in significant short-term morbidity and long-term problems, most notably the development of a therapy-related hematopoietic neoplasm. Therapy-related malignancies usually have a poor outcome, and because of this, there is an effort to elucidate the genetic, molecular, and environmental etiology of these diseases. In this paper, the latest research focused on characterizing therapy-related hematopoietic neoplasms and their pathogenetic mechanisms will be presented along with a compilation of the most common cytogenetic and molecular changes associated with these conditions.
Clinics and Pathology
Etiology
Therapy-related acute lymphoblastic leukemia (t-ALL) is less well characterized than its myeloid counterpart and has a longer latency period (median 7 years) except for those with KMT2A rearrangement, but is otherwise similar in presentation including prior exposure to both cytotoxic and topoisomerase II inhibitors, similar abnormalities of chromosomes 5 and 7, and an inferior outcome [Aldoss et al, 2018].
Epidemiology
With better treatments for cancer, survival rates among many cancers is increasing; however, this will likely result in an increased incidence of t-MN as a long-term complication of this success. Presently, chemotherapy increases the risk of t-MN by 4.7-fold as does younger age at the time of exposure. For breast cancer, the use of hematopoietic growth factors, specifically GSF3 (granulocyte colony-stimulating factor G-CSF) to support blood count recovery also increases the relative risk of t-MN. Changes in treatment protocols have led to changes in incidence of t-MN. For Hodgkin lymphoma, the historical treatment plan included alkylating agent-based chemotherapy with or without extended field radiotherapy, resulting in a 2-6.7% incidence of t-MN, with higher rates if maintenance oral-alkylating agent therapy is utilized; however, with modern therapy protocols including limited radiation, the incidence of t-MN has fallen to 0-0.03% after ten year follow-up. In contrast, risk of t-MN has increased for non-Hodgkin lymphoma, especially following allogenic hematopoietic stem cell transplantation [Churpek et al, 2013; McNerney et al, 2017; Vardiman et al, 2017]. Therapy- related myeloid disorders can also occur as the result of treatments for non-malignant conditions such as autoimmune diseases [Zhang and Wang, 2014], after solid organ transplantation [Dharnidharka 2018], and following autologous or allogenic stem cell transplantation for either malignant or non-malignant conditions [Danylesko and Shimoni, 2018; Saygin et al, 2019].
Therapy-related acute lymphoblastic leukemia (t-ALL) is relatively rare, and for this reason has not been well characterized nor is it a recognized WHO entity in the latest revision. When compared with de novo ALL, t-ALL patients are generally older and more often female. Agents used for immunosuppression to treat autoimmune diseases such as rheumatoid arthritis and following solid organ transplantation and stem cell transplantation also increase the risk of therapy-related lymphoproliferations such as lymphoma [Bagg A, 2011; Aldoss I et al, 2018].
T-ALL has not been as well characterized as its myeloid counterpart because of the lack of large data sets, and so, unlike t-MN, t-ALL is not yet considered a specific entity in the WHO Classification of Tumors of Haematopoietic and Lymphoid Tissue. The frequency of t-ALL is reported to be 3-9% of all adult ALL cases, compared with 10-20% t-MN among all myeloid neoplasms. As with t-MN, the prognosis of t-ALL is significantly worse than in de novo ALL. Comparable is the latency period from cytotoxic therapy to development of t-ALL, with a shorter period associated with topoisomerase II inhibitors and KMT2A rearrangement as compared with other chemotherapy and radiation. The most common primary malignancies associated with t-ALL include breast cancer (26%), plasma cell myeloma (19%), prostate cancer (13%), and non-Hodgkin lymphoma (11%) [Saygin et al, 2019]. The incidence of t-ALL appears to be higher in females, likely due to the high incidence of breast cancer and the multimodality treatment such patients receive, including chemotherapy, adjuvant chemotherapy, and radiation, resulting in long term survival for many.
Because fewer cases of t-ALL have been reported, no large-scale studies examining the pathogenesis of the disease have been published; however, the causes of t-ALL are thought to be like those of t-MN. The majority of cases are believed to be due, in large part, to prior cytotoxic therapy for a primary malignancy which results in DNA damage. Likely also contributing to the pathogenesis of t-ALL, like t-MN, is inherited predisposition to mutations in DNA repair genes such as BRCA1, BRCA2, TP53, CHEK2, and PALB2. There are also ALL patients with a prior malignancy but without any cytotoxic therapy, suggesting the possibility of two primary malignancies without inherited predisposition [Aldos et al, 2019].
Pathology
Other features
The effects of cytotoxic therapy on the bone marrow niche are complex and not completely understood, but have been shown to include a pro-inflammatory response with release of cytokines, damage to HSCs by reactive oxygen species released by multipotent stem cells, and remodelling of the bone marrow niche. Studies in both mice and humans have demonstrated that such exposure to cytotoxic agents creates a "malignant" bone marrow microenvironment that supports selection of mutant HSCs at the expense of healthy HSCs. These mutant stem cells often have reduced TP53 function and apoptosis. Clonal expansion then results in a therapy-related malignancy which, not surprisingly, is difficult to treat. [McNerney et al, 2017].
Cytogenetics
Chromosome 7q contains three MDRs at 7q22, 7q34, and 7q35-q36. Important genes in these regions include the tumor suppressor gene cut-like homeobox 1 (CUX1) at 7q22; pre-mRNA splicing factor LUC7 like 2 (LUC7L2) gene on 7q34; and KMT2C (MLL3) tumor suppressor, enhancer of zeste homolog 2 (EZH2), and cullin 1 (CUL1) genes at 7q35-q36. In addition, an inherited mutation of the sterile alpha motif domain containing protein 9 like (SAMD9L) gene on 7q21.2 predisposes to AML with monosomy 7 [McNerney et al, 2017].
Thirty percent of t-MN patients present balanced chromosome rearrangements, with 10-15% of these cases demonstrating favorable-risk cytogenetic markers t(8;21)(q22;q22), t(15;17) (q22;q21), and less commonly inv(16)(p13q22). In t-MN, while the prognosis associated with these favorable-risk abnormalities is not as good as in de novo AML, it is better than that associated with high-risk markers including del(5q)/monosomy 5, del(7q)/monosomy 7, complex karyotypes, and TP53 gene deletion/mutation. Normal karyotypes are also found in t-MN, although at a lower frequency compared with de novo AML [Vardiman et al, 2017]. Samra et al (2020) explored the prognostic significance of a normal karyotype in t-AML and found that it was associated with an inferior outcome compared with de novo AML; however, this appeared to be largely due to an increased risk of death in remission as opposed to differences in molecular features or relapse risk. Kim et al (2018) found that t-MN patients with a normal karyotype demonstrated better overall survival after allogenic stem cell transplantation compared with patients that possessed other intermediate-risk markers.
Genes
In the study by Ok and colleagues (2015), distinct differences between de novo MN and t-MN included the higher incidence of mutations in TP53, KIT, WT1, PTPN11, IDH1/2, and EZH2 genes in t-MN, with de novo disease demonstrating a greater number of mutations in NPM1 and FLT3. [Ok CY et al, 2015; Vardiman et al, 2016]. Singhal et al (2019) compared the clinical and mutational characteristics of t-MN and primary MDS and found both similarities and differences in mutational signatures as well as clinical outcomes between t-MN and primary MDS. While the mutation frequency was found to be similar between the two entities, the mutation pattern was different, with TP53 and SRSF2 mutations more frequent in t-MN possibly due to clonal selection and expansion of hematopoietic stem cells with these mutations under genotoxic stress.
Kuzmanovic and colleagues (2019) described two genetic pathways leading to t-MN, one with mutation of the TP53 gene occurring in up to 37% of t-MN and the other associated with chromosome 7 abnormalities and activation of the RAS pathway. Both t-MN and de novo AML with mutated TP53 as the initiating event in many cases, often present the del(5q) chromosome/monosomy 5 (often as part of a complex karyotype) and predict a poorer outcome. [McNerney et al, 2017]. Unlike in primary disease where most TP53 mutations occur in the DNA binding domain, those identified in t-MN are often associated with nuclear trafficking. Sometimes, acquisition of a TP53 mutation occurs as a secondary event, predicting a high likelihood of leukemic transformation. The second genetic pathway in t-MN, occurring in 49% of patients with a poor outcome, involves del(7q) chromosome/monosomy 7 and mutation of a gene that results in activation of the RAS pathway. Monosomy 7/del(7q) can also be observed with del(5q)/monosomy 5 in some cases of t-MN, often as part of a complex karyotype [McNerney et al, 2017].
Clonal Selection. The concept of clonal hematopoiesis of indeterminate potential (CHIP) is a relatively new one, described by Steensma et al in 2015 as the presence of neoplasia-related mutation(s) in a population of hematopoietic cells without morphological evidence of bone marrow disease. While almost 1% of healthy middle-aged individuals are found to carry a clonal molecular or cytogenetic abnormality, that figure increases in people 50-70 years old, with clonal expansion occurring over time. In one study utilizing error-corrected sequencing (which enables the accurate detection of clonal mutations as rare as 0.0003 variant allele frequency), CHIP was identified in 95% (19/20) of healthy individuals aged 50-60 years [Young et al, 2016]. Mutations commonly identified in CHIP are also found in AML, including in epigenetic modifying genes such as DNMT3A, ASXL1, and TET2, along with TP53. Common cytogenetic abnormalities found in patients with CHIP are recurrent cytogenetic markers in myeloid neoplasia as well, including del(11q), del(13q), del(20q), and trisomy 8. Not surprisingly, these individuals have an increased lifetime risk of developing a hematological malignancy. There is now strong evidence that some patients with a t-MN possess a pre-existing (before chemotherapy or radiation) mutation(s) in hematopoietic stem cells (HSCs). Such cells, under the stress of chemotherapy or radiation may be selected for in the pool of both normal and mutant HSCs so that they predominately repopulate the bone marrow over time. Having been selected for their increased fitness under genotoxic stress, neoplastic cells in t-MN are resistant to treatment [McNerney et al, 2017]. Certain mutations identified in CHIP, such as DNMT3A and TET2, are also observed in MDS patients, suggesting that CHIP progresses to MDS in some individuals [Katagiri et al, 2019]. The study by Kuzmanovic and colleagues revealed evidence that approximately half of TP53-mutated t-MN may be CHIP-related, with cytotoxic therapies accelerating the progression from CHIP to t-MN. They also identified a two-fold increase in TP53 double mutants in t-MN versus in primary myeloid neoplasia [Kuzmanovic et al, 2020]. Soerensen et al (2020) demonstrated that there may be value in evaluating for clonal hematopoiesis in stem cells harvested at leukophoresis [prior to autologous stem cell transplant (ASCT)] using a myeloid NGS panel. Their study demonstrated that identification of low variant allele frequencies, along with aberrant CD7 expression in patients with primary non-myeloid malignancy may indicate an increased risk of developing tMN after ASCT.
Prognosis
Note
Autoimmune diseases (AD) including systemic lupus erythematosus, rheumatoid arthritis, inflammatory bowel disorders, multiple sclerosis, ankylosing spondylitis, and others are associated not only with an increased risk of lymphoproliferative disorders but myeloid malignancies as well. In a SEER population-based study, a significantly increased risk of either AML or MDS of 1.29 and 1.5, respectively, was found in patients with an AD [Anderson et al, 2009]. The risk of developing a myeloid neoplasm in an AD appears to be based on several factors including shared genetic susceptibilities with polymorphisms in DNA-repair and drug metabolizing genes, treatment exposure (especially to thiopurines, alkylating agents, and topoisomerase inhibitors), chronic bone marrow stimulation, and immune surveillance defects. In addition, rheumatic disease may co-occur with or be a prelude to an underlying hematological malignancy. It should be kept in mind, however, that not all myeloid malignancies that appear in patients with AD are "therapy-related" as some myeloid neoplasms appear in certain AD subtypes in the absence of prior therapy [Boddu et al, 2019].
Cytogenetics
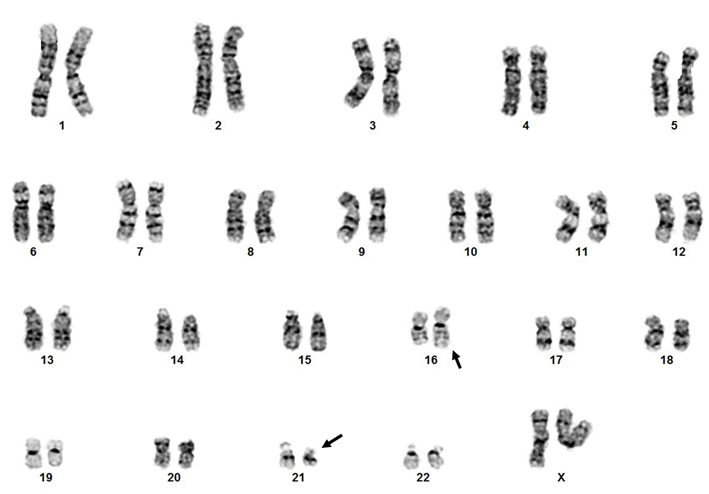
Cytogenetics morphological
- t(4;12)(q12;p13) CHIC2 /ETV6: t-AML and t-ALL; B-cell ALL cases seem to have a more distal breakpoint in 4q13 or 4q21; Median survival is poor, possibly one year.
-t(7;12)(p15;p13), t(12;20)(p13;q11)
- t(12;22)(p13;q12) ETV6/ MN1: t-MN; Survival not certain due to small number of cases, but of nine reported, the survival range was up to 6 years with a median of 2 years.
KMT2A (formally MLL) gene (11q23) rearrangements: t-AML, t-ALL, and biphenotypic acute leukemia; found mainly following treatment with topoisomerase II inhibitors; prior malignancy is variable; prognosis is poor.
- inv(11)(q21q23) KMT2A/ MAML2
- t(1;11)(p32;q23) EPS15 /KMT2A
- t(1;11)(q21;q23) MLLT11 /KMT2A
- t(4;11)(q21;q23) AFF1 /KMT2A
- t(6;11)(q27;q23) AFDN /KMT2A
- t(9;11)(p22;q23) MLLT3 /KMT2A
- t(10;11)(p12;q23) MLLT10 /KMT2A
- t(11;16)(q23;p13.3) KMT2A/ CREBBP
- t(11;17)(q23;q25) KMT2A/ SEPT9
- t(11;19)(q23;p13.3) KMT2A/ MLLT1
- t(11;19)(q23;p13.1) KMT2A/ ELL
RUNX1 (formally AML1) gene (21q22) rearrangements:
- t(8;21)(q22;q22) RUNX1T1 / RUNX1: t-MDS or t-AML; Primary malignancy is solid tumor in 70% of cases; Median survival is 17 months with additional abnormalities and 31 months without additional abnormalities.
- t(3;21)(q26;q22) MECOM /RUNX1: t-MDS and t-AML; Median survival is 8 months.
- t(16;21)(q24;q22) CBFA2T3 /RUNX1: t-AML; Median survival is poor.
RPN1 /MECOM (3q21q26) gene rearrangement:
- inv(3)(q21q26) and t(3;3)(q21;q26): t-MDS or t-AML; primary malignancy is solid tumor in half of cases; Median survival is very poor (7 months).
NUP98 gene (11p15) rearrangements:
- inv(11)(p15q22) and t(11;11)(p15;q22); NUP98/ DDX10: t-MDS and t-AML
- t(7;11)(p15;p15) NUP98/ HOXA9
dic(1;7)(q10;p10) Results in 1q trisomy and 7q monosomy; Half of cases after chemotherapy and radiation; Usually found as the sole abnormality; Breakpoints within α-satellite DNA; Has a distinct gene expression profile associated with gene downregulation supported by a specific epigenetic profile [Fernandez et al, 2019].
t(3;8)(q26.2;q24) MECOM/ MYC rearrangement: Shares similarities with inv(3) GATA2 /MECOM; Dismal outcome.
t(8;16)(p11.2;p13.3) KAT6A / CREBBP rearrangement: Median survival is very poor (5 months); Associated with hemophagocytosis [Xie et al, 2019].
t(9;22)(q34;q11.2) BCR / ABL1 rearrangement: t-AML and t-ALL; primary malignancy is solid tumor in 70% of cases; Median survival is very poor (5 months).
t(15;17)(q22;q21) PML / RARA rearrangement: t-MDS and t-AML; Primary disease is solid tumor in 70% of cases; Median survival is 29 months, although some studies have reported comparable outcomes to de novo acute promyelocytic leukemia [Dhakal et al, 2020].
inv(16)(p13q22) CBFB / MYH11 rearrangement: t-MDS and t-AML; Primary malignancy is solid tumor in 70% of cases; Patients under 55 years appear to have better outcomes; Median survival is 29 months with 45% of patients alive at 5 years (the best survival among subgroups of treatment-related hematopoietic neoplasms with a balanced chromosome rearrangement).
Cytogenetic abnormalities identified in t-ALL are often similar to those observed in t-MN, including monosomal karyotypes; del(5q) and del(7q) chromosomes; KMT2A gene rearrangements; and mutations in genes including DNMT3A, RUNX1, and ASXL1; however, some patients present mutations in genes primarily associated with ALL including CDKN2A IKZF1, FANCL, FANCD2, and BRCA2. The study by Saygin et al, 2019 evaluated patients with t-ALL, de novo ALL (dnALL), and ALL with a prior malignancy but with no history of cytotoxic therapy (pmALL). They found different cytogenetic profiles in t-ALL compared with dnALL and pmALL, with a higher incidence of KMT2A rearrangement (13%) in the t-ALL group compared with 8% in the dnALL group and no cases in the pmALL patients. In addition, 27% of t-ALL patients demonstrated MDS-associated chromosome abnormalities including deletion of chromosomes 5q, 7q, 11q, 13q, 17p, and 20q, along with trisomy 8. By comparison, only 7% of dnALL cases and no cases of pmALL revealed such abnormalities. The t(9;22)(q34;q11.2), the most common abnormality found in adults with de novo ALL, is commonly found in adults with t-ALL as well; however, curiously, 21% of pediatric t-ALL cases in one study presented the Philadelphia chromosome, which is a much higher frequency than what is observed in dnALL in this age group (3%). Cases with the t(9;22) also presented additional chromosome abnormalities at a higher than expected frequency, possibly due to the genetic instability created by prior therapy [Aldoss et al, 2019].
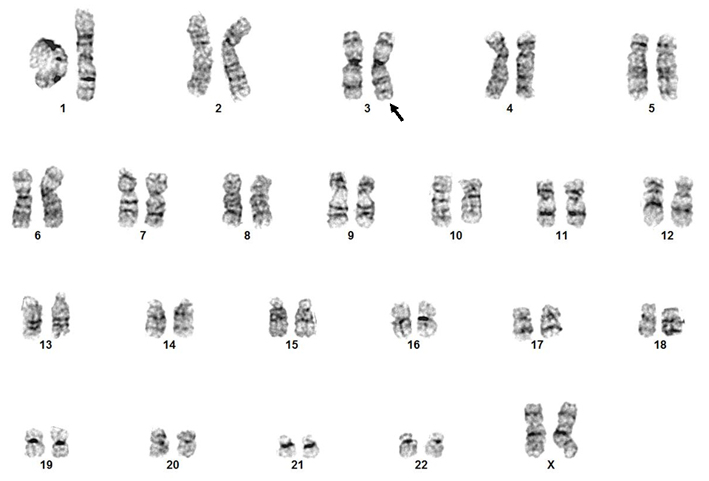
Genes Involved and Proteins
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 31235377 | 2019 | Therapy-related acute lymphoblastic leukemia: Where do we stand with regards to its definition and characterization? | Aldoss I et al |
| 29903756 | 2018 | Therapy-related acute lymphoblastic leukemia has distinct clinical and cytogenetic features compared to de novo acute lymphoblastic leukemia, but outcomes are comparable in transplanted patients. | Aldoss I et al |
| 19259097 | 2009 | Risks of myeloid malignancies in patients with autoimmune conditions. | Anderson LA et al |
| 21490437 | 2011 | Therapy-associated lymphoid proliferations. | Bagg A et al |
| 30927978 | 2019 | Myeloid disorders after autoimmune disease. | Boddu PC et al |
| 30927972 | 2019 | Clinicopathological aspects of therapy-related acute myeloid leukemia and myelodysplastic syndrome. | Chua CC et al |
| 24507808 | 2013 | The evolving challenge of therapy-related myeloid neoplasms. | Churpek JE et al |
| 29423555 | 2018 | Second Malignancies after Hematopoietic Stem Cell Transplantation. | Danylesko I et al |
| 31953046 | 2020 | Treatment Strategies for Therapy-related Acute Myeloid Leukemia. | Dhakal P et al |
| 29178667 | 2018 | Comprehensive review of post-organ transplant hematologic cancers. | Dharnidharka VR et al |
| 18202658 | 2008 | Identification of RPS14 as a 5q- syndrome gene by RNA interference screen. | Ebert BL et al |
| 30923319 | 2019 | A distinct epigenetic program underlies the 1;7 translocation in myelodysplastic syndromes. | Fernandez AGL et al |
| 26304885 | 2015 | Epidemiology and Clinical Significance of Secondary and Therapy-Related Acute Myeloid Leukemia: A National Population-Based Cohort Study. | Granfeldt Østgård LS et al |
| 30927974 | 2019 | Myeloid malignancies after treatment for solid tumours. | Guru Murthy GS et al |
| 31699793 | 2020 | Predisposed genomic instability in pre-treatment bone marrow evolves to therapy-related myeloid neoplasms in malignant lymphoma. | Katagiri S et al |
| 30596716 | 2018 | Better treatment outcomes in patients with actively treated therapy-related myeloid neoplasms harboring a normal karyotype. | Kim SA et al |
| 24429522 | 2014 | Leukaemogenesis induced by an activating β-catenin mutation in osteoblasts. | Kode A et al |
| 31413096 | 2020 | Genomics of therapy-related myeloid neoplasms. | Kuzmanovic T et al |
| 29269520 | 2018 | Emergence and evolution of TP53 mutations are key features of disease progression in myelodysplastic patients with lower-risk del(5q) treated with lenalidomide. | Lodé L et al |
| 28835720 | 2017 | Therapy-related myeloid neoplasms: when genetics and environment collide. | McNerney ME et al |
| 22965552 | 2013 | Transcription factors Fli1 and EKLF in the differentiation of megakaryocytic and erythroid progenitor in 5q- syndrome and in Diamond-Blackfan anemia. | Neuwirtova R et al |
| 25573287 | 2015 | Mutational profiling of therapy-related myelodysplastic syndromes and acute myeloid leukemia by next generation sequencing, a comparison with de novo diseases. | Ok CY et al |
| 32044376 | 2020 | Clonal hematopoiesis in cancer. | Park SJ et al |
| 32366832 | 2020 | Characteristics and outcomes of patients with therapy-related acute myeloid leukemia with normal karyotype. | Samra B et al |
| 31869410 | 2019 | Therapy-related acute lymphoblastic leukemia is a distinct entity with adverse genetic features and clinical outcomes. | Saygin C et al |
| 22652532 | 2012 | Germline mutations in the DNA damage response genes BRCA1, BRCA2, BARD1 and TP53 in patients with therapy related myeloid neoplasms. | Schulz E et al |
| 31089247 | 2019 | The mutational burden of therapy-related myeloid neoplasms is similar to primary myelodysplastic syndrome but has a distinctive distribution. | Singhal D et al |
| 32150606 | 2020 | Clonal hematopoiesis predicts development of therapy-related myeloid neoplasms post-autologous stem cell transplantation. | Soerensen JF et al |
| 25931582 | 2015 | Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. | Steensma DP et al |
| 27218649 | 2016 | The 5q deletion size in myeloid malignancies is correlated to additional chromosomal aberrations and to TP53 mutations. | Stengel A et al |
| 30927971 | 2019 | Germline polymorphisms and the risk of therapy-related myeloid neoplasms. | Takahashi K et al |
| 25487151 | 2015 | Role of TP53 mutations in the origin and evolution of therapy-related acute myeloid leukaemia. | Wong TN et al |
| 30759270 | 2019 | Acute myeloid leukemia with t(8;16)(p11.2;p13.3)/KAT6A-CREBBP in adults. | Xie W et al |
| 27546487 | 2016 | Clonal haematopoiesis harbouring AML-associated mutations is ubiquitous in healthy adults. | Young AL et al |
| 25120730 | 2014 | A focused review of hematopoietic neoplasms occurring in the therapy-related setting. | Zhang L et al |
Citation
Mark A Micale
Therapy-Related Hematopoietic Neoplasia
Atlas Genet Cytogenet Oncol Haematol. 2020-11-01
Online version: http://atlasgeneticsoncology.org/haematological/1303/therapy-related-hematopoietic-neoplasia
