Identity
DNA/RNA
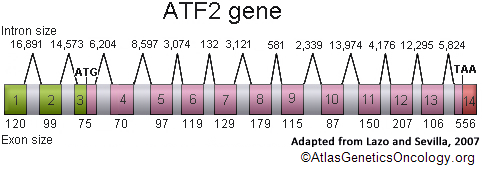
Description
Transcription
Proteins
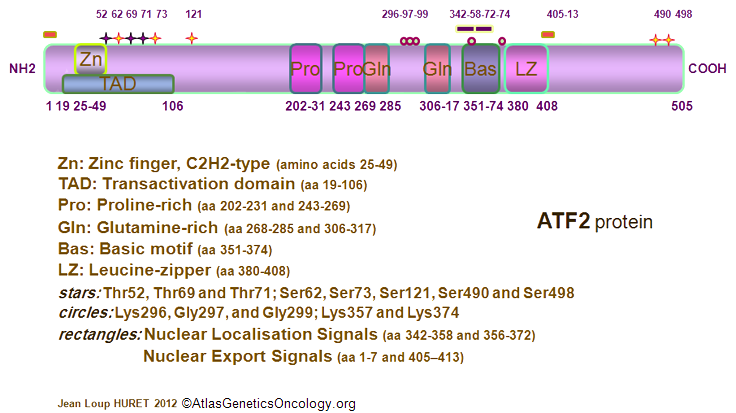
Description
Expression
Localisation
Function
- Typically, it binds to the cAMP-responsive element (CRE) (consensus: 5-GTGACGT[AC][AG]-3), a sequence present in many cellular promoters (Hai et al.,1989). However, depending on the heterodimeric partner, ATF2 binds to different response elements on target genes. ATF2/JUN and ATF2/CREB1 bind the above noted concensus sequence. ATF2 is mainly a transcription activator, but it also may be a transcription repressor (reviews in Bhoumik and Ronai, 2008; Lopez-Bergami et al., 2010; Lau and Ronai, 2012).
Activation of ATF2
ATF2 is activated by stress kinases, including JNK (MAPK8, MAPK9, MAPK10) and p38 (MAPK1, MAPK11, MAPK12, MAPK13, MAPK14) and is implicated in transcriptional regulation of immediate early genes regulating stress and DNA damage responses (Gupta et al., 1995; van Dam et al., 1995) and cell cycle control under normal growth conditions.(up-regulation of the CCNA2 (cyclin A) promoter at the G1/S boundary) (Nakamura et al., 1995). In response to stimuli, ATF2 is phosphorylated on threonine 69 and/or 71 by JNK or by p38. Phosphorylation on Thr69 and Thr71 of ATF2 and its dimerization are required to activate ATF2 transcription factor activity. Phosphorylation on Thr69 occurs through the RALGDS-SRC-P38 pathway, and phosphorylation on Thr71 occurs through the RAS-MEK-ERK pathway (MAPK1, MAPK3 (ERK1), MAPK11, MAPK12 and MAPK14) (Gupta et al., 1995; Ouwens et al., 2002).
The intrinsic histone acetylase activity of ATF2 promotes its DNA binding ability (Abdel-Hafiz et al., 1992; Kawasaki et al., 2000).
Interaction of ATF2 with CREBBP (CREB-binding protein, also called p300/CBP) is dependent upon phosphorylation at Ser121 induced by PRKCA. ATF2 and CREBBP cooperate in the activation of transcription (Kawasaki et al., 1998; Yamasaki et al., 2009).
VRK1 activates and stabilizes ATF2 through direct phosphorylation of Ser62 and Thr73 (Sevilla et al., 2004).
Down regulation of ATF2
Among other down regulation mechanisms, ATF2 is down regulated, by MIR26B in response to ionizing radiation (Arora et al., 2011).
Transcriptionally active dimers of ATF2 protein are regulated by ubiquitylation and proteosomal degradation (Fuchs et al., 1999); phosphorylation of ATF2 on Thr69 and Thr71 promotes its ubiquitylation and degradation (Firestein and Feuerstein, 1998).
A cytoplasmic alternatively spliced isoform of ATF7, ATF7-4, is a cytoplasmic negative regulator of both ATF2 and ATF7. It impairs ATF2 and ATF7 phosphorylation (ATF7-4 indeed sequesters the Thr53-phosphorylating kinase in the cytoplasm, preventing Thr53 phosphorylation of ATF7) and transcriptional activity (Diring et al., 2011).
The activity of ATF2 is repressed by an intramolecular interaction between the N-terminal domain and the b-ZIP domain (Li and Green, 1996). The N-terminal nuclear export signal (NES) of ATF2 negatively regulates ATF2 transcriptional activity (Hsu and Hu, 2012).
Dimerization of ATF2
The basic leucine zipper (basic motif + leucine zipper, "b-ZIP") of ATF2 enables homo- or hetero-dimerization.
The main dimerization partners of ATF2 are the following: ATF2, BRCA1, CREB1, JDP2, JUN, JUNB, JUND, MAFA, NF1, NFYA, PDX1, POU2F1, TCF3 (Lau and Ronai, 2012).
ATF2 homodimers have a low transcriptional activity.
MAPKAP1 (SIN1) binds to the b-ZIP region of ATF2, and also binds MAPK14, and is required for MAPK14-induced phosphorylation of ATF2 in response to osmotic stress, and activates the transcription of apoptosis-related genes (Makino et al., 2006).
In response to stress, ATF2 binds to POU2F1 (OCT1), NFI, and BRCA1 to activate transcription of GADD45. The b-Zip region of ATF2 is critical for binding to BRCA1. ATF2 also binds and activates SERPINB5 (Maspin) (Maekawa et al., 2008).
ATF2 also forms a heterodimer with JDP2, a repressor of AP-1. JDP2 inhibits the transactivation of JUN by ATF2 (Jin et al., 2002).
ATF2 target genes
Under genotoxic stress, a study showed that 269 genes were found to be bound by ATF2/JUN dimers. Immediate-early genes were a notable subset and included EGR family members, FOS family members, and JUN family members, but the largest group of genes belonged to the DNA repair machinery (Hayakawa et al., 2004, see below).
Among the ATF2 target genes are :
- Transcription factors, such as JUN, ATF3, DDIT3 (CHOP), FOS, JUNB,
- DNA damage proteins (see below),
- Cell cycle regulators (CCNA2, CCND1), see below,
- Regulators of apoptosis (see below and Hayakawa et al., 2004),
- Growth factor receptors and cytokines such as PDGFRA (Maekawa et al., 1999), IL8 (Agelopoulos and Thanos, 2006), FASLG (Fas ligand), TNF (TNFalpha), TNFSF10 family (Herr et al., 2000; Faris et al., 1998),
- Proteins related with invasion such as MMP2 (Hamsa and Kuttan, 2012) and PLAU (UPA): ATF2/JUN heterodimer binds to and activates PLAU (De Cesare et al., 1995),
- Cell adhesion molecules, such as SELE, SELP, and VCAM1 (Reimold et al., 2001),
- Proteins engaged in the response to endoplasmic reticulum (ER) stress. ATF2/CREB dimers bind the CRE-like element TGACGTGA of HSPA5 (Grp78) and activates it (Chen et al., 1997),
- Genes encoding extracellular matrix components seem to constitute an important subset of ATF2/JUN-target genes (van Dam and Castellazzi, 2001).
- PTEN, a negative regulator of the PI3K/AKT signaling pathway, is positively regulated by ATF2 (Qian et al., 2012).
- ATF1 and ATF2 regulate TCRA and TCRB gene transcription.
Histones, Chromatin
UV treatment or ATF2 phosphorylation increases its histone acetyltransferase (HAT) activity as well as its transcriptional activities. Lys296, Gly297 and Gly299, are essential both for histone acetyltransferase activity and for transactivation (Kawasaki et al., 2000).
Binding of ATF2 to the histone acetyltransferase RUVBL2 (TIP49b) suppresses ATF2 transcriptional activity. RUVBL2s association with ATF2 is phosphorylation dependent and requires amino acids 150 to 248 of ATF2 (Cho et al., 2001).
ATF2 interacts with the acetyltransferase domains of CREBBP. ATF2 b-ZIP could serve as an acetyltransferase substrate for the acetyltransferase domains of CREBBP. ATF2 is acetylated on Lys357 and Lys374 by CREBBP, which contributes to its transcriptional activity (Karanam et al., 2007).
ATF2 and ATF4 are essential for the transcriptional activation of DDIT3 (CHOP) upon amino acid starvation.
ATF2 is essential in the acetylation of histone H4 and H2B, and thereby may be involved in the modification of the chromatin structure. An ATF2-independent HAT activity is involved in the amino acid regulation of ASNS transcription (Bruhat et al., 2007).
The histone variant macroH2A recruitement into nucleosomes could confer an epigenetic mark for gene repression. The constitutive DNA binding of the ATF2/JUND heterodimer to the IL-8 enhancer recruits macroH2A-containing nucleosomes in B cells, thus inhibiting transcriptional activation (Agelopoulos and Thanos, 2006).
Heat shock or osmotic stress induces phosphorylation of dATF2 (ATF2 in Drosophila), results in its release from heterochromatin, and heterochromatic disruption. dATF2 regulates heterochromatin formation. ATF2 may be involved in the epigenetic silencing of target genes in euchromatin. The stress-induced ATF2-dependent epigenetic change was maintained over generations, suggesting a mechanism by which the effects of stress can be inherited (Seong et al., 2011).
DNA damage response
Phosphorylation on Ser490 and Ser498 by ATM is required for the activation of ATF2 in DNA damage response. Phosphorylation of ATF2 results in the localisation of ATF2 in ionizing radiation induced foci (in cells exposed to ionizing radiation (IR), several proteins phosphorylated by ATM translocate and colocalize to common intranuclear sites. The resulting IR-induced nuclear foci (IRIF) accumulate at the sites of DNA damage). ATF2 expression contributes to the selective recruitment of MRE11A, RAD50, and NBN (NBS1) into IRIF. ATF2 is required for the IR-induced S phase checkpoint, and this function is independant of its transcriptional activity (Bhoumik et al., 2005).
KAT5 (TIP60) is a histone acetyltransferase and chromatin-modifying protein involved in double strand breaks (DSB) repair, interacting with and acetylating ATM. ATF2 associates with KAT5 and RUVBL2. Under non-stressed conditions, ATF2 in cooperation with the ubiquitin ligase CUL3 promotes the degradation of KAT5 (Bhoumik et al., 2008a).
Following genotoxic stress, 269 genes were found to be bound by ATF2/JUN dimers (see above), of which were 23 DNA repair or repair-associated genes (ERCC1, ERCC3, XPA, MSH2, MSH6, RAD50, RAD23B, MLH1, HIST1H2AC, PMS2, FOXN3 (CHES1), LIG1, ERCC8 (CKN1), UNG, XRCC6 (G22P1), TREX1, PNP, GTF2H1, ATM, FOXD1, DDX3X, DMC1, and the DNA repair-associated GADD45G), derived from several recognized DNA repair mechanisms (Hayakawa et al., 2004).
Cell Cycle
RB1 constrains cellular proliferation by activating the expression of the inhibitory growth factor TGFB2 (TGF-beta 2) through ATF2 (Kim et al., 1992).
CREB1 dimerizes with ATF2 to bind to the CCND1 (cyclin D1) promoter, to increase CCD1 expression (Beier et al., 1999).
JUND dimerizes with ATF2 to repress CDK4 transcription, a protein necessary for the G1-to-S phase transition during the cell cycle, by binding to the proximal region of the CDK4-promoter, contributing to the inhibition of cell growth. The physical interactions of ATF2 with JUND implicates the b-ZIP domain of ATF2 (Xiao et al., 2010).
Heterodimerization of JUND with ATF2 activates CCNA2 (cyclin A) promoter. CCNA2 is essential for the control at the G1/S and the G2/M transitions of the cell cycle. In contrast, ATF4 expression suppresses the promoter activation mediated by ATF2 (Shimizu et al., 1998).
Apoptosis
ATF2/CREB1 heterodimer binds to the CRE element of the BCL2 promoter (Ma et al., 2007).
ATF2 induces BAK upregulation (Chen et al., 2010).
MAP3K5 (ASK1) activates ATF2 and FADD-CASP8-BID signalling, resulting in the translocation of BAX and BAK, and subsequently mitochondrial dysregulation (Hassan et al., 2009).
ATF2/JUN heterodimers bind and activate CASP3, a key executor of neuronal apoptosis (Song et al., 2011).
Following death receptor stimulation, there is phosphorylation and binding of ATF2/JUN to death-inducing ligands promoters (FASLG, TNF, TNFSF10), which allows the spread of death signals (Herr et al., 2000). Neuronal apoptosis requires the concomitant activation of ATF2/JUN and downregulation of FOS (Yuan et al., 2009).
Many drugs are currently being tested for their ability to inhibit cell proliferation and induce apoptosis through various pathways, including ATF2 pathway.
In the cytoplasm, ATF2 abrogates formation of complexes containing HK1 and VDAC1, deregulating mitochondrial outer-membrane permeability and initiating apoptosis. This function is negatively regulated phosphorylation of ATF2 by PRKCE, which dictates its nuclear localization (Lau et al., 2012).
Metabolic control and Insulin signalling
ATF2 has been implicated in the regulation of proteins involved in metabolic control, including the control of the expression of UCP1, a protein involved in thermogenic response in brown adipose tissue (Cao et al., 2004) and phosphoenolpyruvate carboxykinase (PEPCK), a protein regulating gluconeogenesis (Cheong et al., 1998).
Insulin activates ATF2 by phosphorylation of Thr69 and Thr71 (Baan et al., 2006).
Co-expression of ATF2, MAFA, PDX1, and TCF3 results in a synergistic activation of the insulin promoter in endocrine cells of pancreatic islets. ATF2, MAFA, PDX1, and TCF3 form a multi-protein complex to facilitate insulin gene transcription (Han et al., 2011).
ATF2 target genes in insulin signalling are ATF3, JUN, EGR1, DUSP1 (MKP1), and SREBF1. Deregulation of these genes is linked to the pathogenesis of insulin resistance, beta-cell dysfunction and vascular complications found in type 2 diabetes. Therefore, aberrant ATF2 activation under conditions of insulin resistance may contribute to the development of type 2 diabetes (Baan et al., manuscript in preparation).
Iron depletion
Iron depletion induced by chelators increases the phosphorylation of JNK and MAPK14, as well as the phosphorylation of their downstream targets p53 and ATF2 (Yu and Richardson, 2011).
Mutations
Note
Somatic
Implicated in
ATF2/CREB1 binds to CRE domain of TNFSF11 (RANKL) promoter and TNFSF11 expression stimulates osteoclastogenesis in mouse stromal/osteoblast cells (Bai et al., 2005). Binding of JUN CREB1, ATF1, and ATF2 complexes are required for COL24A1 transcription, a marker of late osteoblast differentiation (Matsuo et al., 2006). Luteolin, a flavonoid, inhibits TNFSF11-induced osteoclastogenesis through the inhibition of ATF2 phosphorylation (Lee et al., 2009).
Altogether, ATF2 seems to play a fundamental role in neuronal viability and in neurological functions in the normal brain and is down-regulated in the hippocampus and the caudate nucleus in Alzheimer, Parkinson and Huntington diseases (Pearson et al., 2005).
Mice carrying a germline mutation in ATF2 had a reduced number of cerebellar Purkinje cells, atrophic vestibular sense organs, an ataxic gait, hyperactivity,and decreased hearing (Reimold et al., 1996). A missense mutation in ATF2 in dogs has shown to provoke an autosomal recessive disease with short stature and weakness at birth, ataxia and generalized seizures, dysplastic foci consisting of clusters of intermixed granule and Purkinje cells, and death before 7 weeks of age (Chen et al., 2008d).
ATF2 plays critical roles for the expression of the TH gene (tyrosine hydroxylase) and for neurite extension of catecholaminergic neurons (Kojima et al., 2008).
Neuronal-specific ATF2 expression is required for embryonic survival, ATF2 has a strong pro-survival role in somatic motoneurons of the brainstem, and loss of functional ATF2 leads to hyperphosphorylated JNK and p38, and results in somatic and visceral motoneuron degeneration (Ackermann et al., 2011).
On the other hand, activated ATF2 promotes apoptosis of various brain cells, of which are cerebellar granule neurons (Ramiro-Cortés et al., 2011; Song et al., 2011). ATF2/JUN heterodimers bind and activate CASP3, a key executor of neuronal apoptosis, in cerebellar granule neurons (Song et al., 2011).
ATF2/JUN heterodimers bind and activate HRK (DP5, death protein 5/harakiri), a proapoptotic gene, promoting the death of sympathetic neurons (Ma et al., 2007; Towers et al., 2009), but also ATF2/JUN binds to two conserved CRE sites in the DUSP1 (MKP1) promoter; overexpression of DUSP1 inhibits JNK-mediated phosphorylation of JUN and protect sympathetic neurons from apoptosis (Kristiansen et al., 2010).
ATF2 overexpression in nucleus accumbens produces increases in emotional reactivity and antidepressant-like responses (Green et al., 2008), see Table 1.

ATF2 attenuates melanoma susceptibility to apoptosis. ATF2 control of melanoma development is mediated through its negative regulation of SOX10 and consequently of MITF transcription. The ratio of nuclear ATF2 to MITF expression is associated with poor prognosis (Shah et al., 2010). Assignment to the low-risk group in stage II melanoma requires elevated levels of overall CTNNB1 (beta-catenin) and nuclear CDKN1A (p21WAF1), decreased levels of fibronectin, and distributions that favor nuclear concentration for CDKN2A (p16INK4A) but cytoplasmic concentration for ATF2 (Gould Rothberg et al., 2009).
Phosphorylated ATF2 (p-ATF2) is significantly overexpressed in cutaneous angiosarcoma (malignant tumor) and pyogenic granuloma (benign tumor) than in normal dermal vessels (Chen et al., 2008a); p-ATF2 is also overexpressed in cutaneous squamous cell carcinoma, Bowens disease, and basal cell carcinomas, as compared to its expression in normal skin (Chen et al., 2008b).
High levels of ATF2/JUN dimers induce autocrine growth and primary tumor formation of fibrosarcomas in the chicken (van Dam and Castellazzi, 2001). ATF2 mutant mice in which the ATM phosphoacceptor sites (S472/S480) were mutated (ATF2KI mice) are more sensitive to ionizing radiation IR, exhibit increased intestinal cell apoptosis, develop a higher number of low-grade skin tumors (papillomas, squamous cell carcinomas, spindle cell carcinomas) (Li et al., 2010). A decrease of nuclear ATF2 and high CTNNB1 (beta-catenin) expression is seen in squamous cell carcinoma and basal cell carcinoma, compared to normal skin, while the cytoplasmic ATF2 expression was not significantly different in cancer and normal skin (Bhoumik et al., 2008b).
A nuclear localization of ATF2 would be associated with its oncogenic properties, and a cytosolic localization with its tumor suppressor properties (Lau and Ronai, 2012).
NFE2L2 (NRF2) is a transcription activator of the bZIP family which binds to antioxidant response elements (ARE) in the promoter regions of target genes in response to oxidative stress. NFE2L2 positively regulates the expression the AP-1 family proteins ATF2, JUN and FOS. NFE2L2/ARE pathway plays an important role in the induction of differentiation of myeloid leukemia cells by 1alpha,25-dihydroxyvitamin D3 (1,25D), a strong differentiation agent (Bobilev et al., 2011). A crosstalk between NFE2L2 and ATF2 has also been noted in prostate cancer cells (Nair et al., 2010).
ATF2/JUN mediate increased BIM expression in response to MAPK14 (p38alpha) signaling in cells detached from the extracellular matrix, indicating a contributing role for ATF2 in regulating acinar lumen formation, crucial for the development of mammary gland development, a function that may be crucial to its ability to suppress breast cancer. (Wen et al., 2011).
ATF2/JUN binds to a potential CRE element of FOXP3, and induces its expression. FOXP3 acts as a transcriptional repressor of oncogenes such as ERBB2 and SKP2, and is able to cause apoptosis of breast cancer cells. The use of this ATF2-FOXP3 pathway may be of potential interest in future therapeutic approach of breast cancer. (Liu et al., 2009).
Aggressive basal-like breast cancer cells exhibit high expression of FOSL1 (FRA1)/JUN dimers rather than ATF2/JUN dimers (Baan et al., 2010).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 21533046 | 2011 | Loss of ATF2 function leads to cranial motoneuron degeneration during embryonic mouse development. | Ackermann J et al |
| 17036053 | 2006 | Epigenetic determination of a cell-specific gene expression program by ATF-2 and the histone variant macroH2A. | Agelopoulos M et al |
| 18802055 | 2008 | Promoter analysis reveals critical roles for SMAD-3 and ATF-2 in expression of IL-23 p19 in macrophages. | Al-Salleeh F et al |
| 21901137 | 2011 | Coordinated regulation of ATF2 by miR-26b in γ-irradiated lung cancer cells. | Arora H et al |
| 16601071 | 2006 | The role of c-Jun N-terminal kinase, p38, and extracellular signal-regulated kinase in insulin-induced Thr69 and Thr71 phosphorylation of activating transcription factor 2. | Baan B et al |
| 15731115 | 2005 | Reactive oxygen species stimulates receptor activator of NF-kappaB ligand expression in osteoblast. | Bai XC et al |
| 15691875 | 2005 | Identification of human myometrial target genes of the c-Jun NH2-terminal kinase (JNK) pathway: the role of activating transcription factor 2 (ATF2) and a novel spliced isoform ATF2-small. | Bailey J et al |
| 11932306 | 2002 | Characterization and functional analysis of cAMP response element modulator protein and activating transcription factor 2 (ATF2) isoforms in the human myometrium during pregnancy and labor: identification of a novel ATF2 species with potent transactivation properties. | Bailey J et al |
| 12118074 | 2002 | p38 mitogen-activated protein kinase is required for TGFbeta-mediated fibroblastic transdifferentiation and cell migration. | Bakin AV et al |
| 9990041 | 1999 | Identification of the cyclin D1 gene as a target of activating transcription factor 2 in chondrocytes. | Beier F et al |
| 18227516 | 2008 | Suppressor role of activating transcription factor 2 (ATF2) in skin cancer. | Bhoumik A et al |
| 11234888 | 2001 | Activating transcription factor 2-derived peptides alter resistance of human tumor cell lines to ultraviolet irradiation and chemical treatment. | Bhoumik A et al |
| 15010535 | 2004 | Transcriptional switch by activating transcription factor 2-derived peptide sensitizes melanoma cells to apoptosis and inhibits their tumorigenicity. | Bhoumik A et al |
| 17935492 | 2007 | ATF2 on the double - activating transcription factor and DNA damage response protein. | Bhoumik A et al |
| 18677098 | 2008 | ATF2: a transcription factor that elicits oncogenic or tumor suppressor activities. | Bhoumik A et al |
| 18397884 | 2008 | Regulation of TIP60 by ATF2 modulates ATM activation. | Bhoumik A et al |
| 15916964 | 2005 | ATM-dependent phosphorylation of ATF2 is required for the DNA damage response. | Bhoumik A et al |
| 22082005 | 2012 | Induction of group IVC phospholipase A2 in allergic asthma: transcriptional regulation by TNFα in bronchoepithelial cells. | Bickford JS et al |
| 21099366 | 2011 | The Nrf2 transcription factor is a positive regulator of myeloid differentiation of acute myeloid leukemia cells. | Bobilev I et al |
| 17267404 | 2007 | ATF2 is required for amino acid-regulated transcription by orchestrating specific histone acetylation. | Bruhat A et al |
| 15024092 | 2004 | p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. | Cao W et al |
| 19670268 | 2009 | JNK1/c-Jun and p38 alpha MAPK/ATF-2 pathways are responsible for upregulation of Fas/FasL in human chronic myeloid leukemia K562 cells upon exposure to Taiwan cobra phospholipase A2. | Chen KC et al |
| 19780038 | 2010 | Calcium-stimulated mitogen-activated protein kinase activation elicits Bcl-xL downregulation and Bak upregulation in notexin-treated human neuroblastoma SK-N-SH cells. | Chen KC et al |
| 9310369 | 1997 | Rapid induction of the Grp78 gene by cooperative actions of okadaic acid and heat-shock in 9L rat brain tumor cells--involvement of a cAMP responsive element-like promoter sequence and a protein kinase A signaling pathway. | Chen KD et al |
| 17869487 | 2008 | Overexpression of phosphorylated ATF2 and STAT3 in eccrine porocarcinoma and eccrine poroma. | Chen SY et al |
| 18074159 | 2008 | A neonatal encephalopathy with seizures in standard poodle dogs with a missense mutation in the canine ortholog of ATF2. | Chen X et al |
| 9712902 | 1998 | Activating transcription factor-2 regulates phosphoenolpyruvate carboxykinase transcription through a stress-inducible mitogen-activated protein kinase pathway. | Cheong J et al |
| 11713276 | 2001 | TIP49b, a regulator of activating transcription factor 2 response to stress and DNA damage. | Cho SG et al |
| 7624151 | 1995 | Heterodimerization of c-Jun with ATF-2 and c-Fos is required for positive and negative regulation of the human urokinase enhancer. | De Cesare D et al |
| 21858082 | 2011 | A cytoplasmic negative regulator isoform of ATF7 impairs ATF7 and ATF2 phosphorylation and transcriptional activity. | Diring J et al |
| 22345152 | 2012 | Tumor cell-derived angiopoietin-like protein ANGPTL2 is a critical driver of metastasis. | Endo M et al |
| 9710625 | 1998 | Stress-induced Fas ligand expression in T cells is mediated through a MEK kinase 1-regulated response element in the Fas ligand promoter. | Faris M et al |
| 22135748 | 2011 | Role of heparan sulfate 2-o-sulfotransferase in prostate cancer cell proliferation, invasion, and growth factor signaling. | Ferguson BW et al |
| 9488727 | 1998 | Association of activating transcription factor 2 (ATF2) with the ubiquitin-conjugating enzyme hUBC9. Implication of the ubiquitin/proteasome pathway in regulation of ATF2 in T cells. | Firestein R et al |
| 10207054 | 1999 | Ubiquitination and degradation of ATF2 are dimerization dependent. | Fuchs SY et al |
| 10777545 | 2000 | Stability of the ATF2 transcription factor is regulated by phosphorylation and dephosphorylation. | Fuchs SY et al |
| 19884546 | 2009 | Melanoma prognostic model using tissue microarrays and genetic algorithms. | Gould Rothberg BE et al |
| 18305237 | 2008 | Induction of activating transcription factors (ATFs) ATF2, ATF3, and ATF4 in the nucleus accumbens and their regulation of emotional behavior. | Green TA et al |
| 7824938 | 1995 | Transcription factor ATF2 regulation by the JNK signal transduction pathway. | Gupta S et al |
| 1827203 | 1991 | Cross-family dimerization of transcription factors Fos/Jun and ATF/CREB alters DNA binding specificity. | Hai T et al |
| 2516827 | 1989 | Transcription factor ATF cDNA clones: an extensive family of leucine zipper proteins able to selectively form DNA-binding heterodimers. | Hai TW et al |
| 21953764 | 2012 | Berberine inhibits pulmonary metastasis through down-regulation of MMP in metastatic B16F-10 melanoma cells. | Hamsa TP et al |
| 21278380 | 2011 | ATF2 interacts with beta-cell-enriched transcription factors, MafA, Pdx1, and beta2, and activates insulin gene transcription. | Han SI et al |
| 19940360 | 2009 | Fas-induced apoptosis of renal cell carcinoma is mediated by apoptosis signal-regulating kinase 1 via mitochondrial damage-dependent caspase-8 activation. | Hassan M et al |
| 15546613 | 2004 | Identification of promoters bound by c-Jun/ATF2 during rapid large-scale gene activation following genotoxic stress. | Hayakawa J et al |
| 10980599 | 2000 | Autoamplification of apoptosis following ligation of CD95-L, TRAIL and TNF-alpha. | Herr I et al |
| 22275354 | 2012 | Critical role of N-terminal end-localized nuclear export signal in regulation of activating transcription factor 2 (ATF2) subcellular localization and transcriptional activity. | Hsu CC et al |
| 12789291 | 2003 | Death receptors and melanoma resistance to apoptosis. | Ivanov VN et al |
| 12052888 | 2002 | JDP2, a repressor of AP-1, recruits a histone deacetylase 3 complex to inhibit the retinoic acid-induced differentiation of F9 cells. | Jin C et al |
| 22797074 | 2013 | SS18-SSX2 and the mitochondrial apoptosis pathway in mouse and human synovial sarcomas. | Jones KB et al |
| 18772397 | 2008 | Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. | Jones S et al |
| 2320002 | 1990 | A cDNA for a human cyclic AMP response element-binding protein which is distinct from CREB and expressed preferentially in brain. | Kara CJ et al |
| 17590016 | 2007 | Multiple roles for acetylation in the interaction of p300 HAT with ATF-2. | Karanam B et al |
| 9436983 | 1998 | p300 and ATF-2 are components of the DRF complex, which regulates retinoic acid- and E1A-mediated transcription of the c-jun gene in F9 cells. | Kawasaki H et al |
| 1641004 | 1992 | Retinoblastoma gene product activates expression of the human TGF-beta 2 gene through transcription factor ATF-2. | Kim SJ et al |
| 17896792 | 2008 | Increased expression of tyrosine hydroxylase and anomalous neurites in catecholaminergic neurons of ATF-2 null mice. | Kojima M et al |
| 20702711 | 2010 | Mkp1 is a c-Jun target gene that antagonizes JNK-dependent apoptosis in sympathetic neurons. | Kristiansen M et al |
| 22304920 | 2012 | PKCε promotes oncogenic functions of ATF2 in the nucleus while blocking its apoptotic function at mitochondria. | Lau E et al |
| 22685333 | 2012 | ATF2 - at the crossroad of nuclear and cytosolic functions. | Lau E et al |
| 20162352 | 2009 | Inhibitory effect of luteolin on osteoclast differentiation and function. | Lee JW et al |
| 20740050 | 2010 | Radiation Sensitivity and Tumor Susceptibility in ATM Phospho-Mutant ATF2 Mice. | Li S et al |
| 8598283 | 1996 | Intramolecular inhibition of activating transcription factor-2 function by its DNA-binding domain. | Li XY et al |
| 20178980 | 2010 | cAMP/CREB-mediated transcriptional regulation of ectonucleoside triphosphate diphosphohydrolase 1 (CD39) expression. | Liao H et al |
| 17158955 | 2006 | JunB is required for endothelial cell morphogenesis by regulating core-binding factor beta. | Licht AH et al |
| 20562914 | 2010 | Cell transformation by v-Rel reveals distinct roles of AP-1 family members in Rel/NF-kappaB oncogenesis. | Liss AS et al |
| 16511568 | 2006 | Mutual regulation of c-Jun and ATF2 by transcriptional activation and subcellular localization. | Liu H et al |
| 19584270 | 2009 | Activating transcription factor 2 and c-Jun-mediated induction of FoxP3 for experimental therapy of mammary tumor in the mouse. | Liu Y et al |
| 7737129 | 1995 | ATF-2 contains a phosphorylation-dependent transcriptional activation domain. | Livingstone C et al |
| 20029425 | 2010 | Emerging roles of ATF2 and the dynamic AP1 network in cancer. | Lopez-Bergami P et al |
| 17428807 | 2007 | dp5/HRK is a c-Jun target gene and required for apoptosis induced by potassium deprivation in cerebellar granule neurons. | Ma C et al |
| 17219413 | 2007 | Activating transcription factor 2 controls Bcl-2 promoter activity in growth plate chondrocytes. | Ma Q et al |
| 10364225 | 1999 | Mouse ATF-2 null mutants display features of a severe type of meconium aspiration syndrome. | Maekawa T et al |
| 17700520 | 2008 | ATF-2 controls transcription of Maspin and GADD45 alpha genes independently from p53 to suppress mammary tumors. | Maekawa T et al |
| 17054722 | 2006 | Sin1 binds to both ATF-2 and p38 and enhances ATF-2-dependent transcription in an SAPK signaling pathway. | Makino C et al |
| 16373341 | 2006 | CREB-AP1 protein complexes regulate transcription of the collagen XXIV gene (Col24a1) in osteoblasts. | Matsuo N et al |
| 11535812 | 2001 | Distinct effects of cAMP and mitogenic signals on CREB-binding protein recruitment impart specificity to target gene activation via CREB. | Mayr BM et al |
| 10092462 | 1999 | Solution structure of the transactivation domain of ATF-2 comprising a zinc finger-like subdomain and a flexible subdomain. | Nagadoi A et al |
| 20729872 | 2010 | Regulation of Nrf2- and AP-1-mediated gene expression by epigallocatechin-3-gallate and sulforaphane in prostate of Nrf2-knockout or C57BL/6J mice and PC-3 AP-1 human prostate cancer cells. | Nair S et al |
| 7843287 | 1995 | Down-regulation of the cyclin A promoter in differentiating human embryonal carcinoma cells is mediated by depletion of ATF-1 and ATF-2 in the complex at the ATF/CRE site. | Nakamura T et al |
| 12110590 | 2002 | Growth factors can activate ATF2 via a two-step mechanism: phosphorylation of Thr71 through the Ras-MEK-ERK pathway and of Thr69 through RalGDS-Src-p38. | Ouwens DM et al |
| 15878807 | 2005 | Activating transcription factor 2 expression in the adult human brain: association with both neurodegeneration and neurogenesis. | Pearson AG et al |
| 22367504 | 2012 | Regulation of phosphatase and tensin homolog on chromosome 10 in response to hypoxia. | Qian J et al |
| 21683152 | 2011 | Reactive oxygen species participate in the p38-mediated apoptosis induced by potassium deprivation and staurosporine in cerebellar granule neurons. | Ramiro-Cortés Y et al |
| 11157857 | 2001 | Decreased immediate inflammatory gene induction in activating transcription factor-2 mutant mice. | Reimold AM et al |
| 21703449 | 2011 | Inheritance of stress-induced, ATF-2-dependent epigenetic change. | Seong KH et al |
| 15105425 | 2004 | Human vaccinia-related kinase 1 (VRK1) activates the ATF2 transcriptional activity by novel phosphorylation on Thr-73 and Ser-62 and cooperates with JNK. | Sevilla A et al |
| 21203491 | 2010 | A role for ATF2 in regulating MITF and melanoma development. | Shah M et al |
| 9511728 | 1998 | Activation of the rat cyclin A promoter by ATF2 and Jun family members and its suppression by ATF4. | Shimizu M et al |
| 21996423 | 2011 | Caspase-3 is a target gene of c-Jun:ATF2 heterodimers during apoptosis induced by activity deprivation in cerebellar granule neurons. | Song B et al |
| 1829805 | 1991 | Expression of the CRE-BP1 transcriptional regulator binding to the cyclic AMP response element in central nervous system, regenerating liver, and human tumors. | Takeda J et al |
| 19304750 | 2009 | The proapoptotic dp5 gene is a direct target of the MLK-JNK-c-Jun pathway in sympathetic neurons. | Towers E et al |
| 18638549 | 2008 | Activating transcription factor-2 affects skeletal growth by modulating pRb gene expression. | Vale-Cruz DS et al |
| 21980432 | 2011 | Transcriptional networks in epithelial-mesenchymal transition. | Venkov C et al |
| 11836564 | 2002 | Infrequent mutations of the activating transcription factor-2 gene in human lung cancer, neuroblastoma and breast cancer. | Woo IS et al |
| 20181929 | 2010 | Induced ATF-2 represses CDK4 transcription through dimerization with JunD inhibiting intestinal epithelial cell growth after polyamine depletion. | Xiao L et al |
| 20087603 | 2010 | Differential sensitivity of human endometrial carcinoma cells with different PTEN expression to mitogen-activated protein kinase signaling inhibits and implications for therapy. | Xiao L et al |
| 22109562 | 2012 | The effect of JDP2 and ATF2 on the epithelial-mesenchymal transition of human pancreatic cancer cell lines. | Xu Y et al |
| 19176525 | 2009 | Phosphorylation of Activation Transcription Factor-2 at Serine 121 by Protein Kinase C Controls c-Jun-mediated Activation of Transcription. | Yamasaki T et al |
| 21378396 | 2011 | Cellular iron depletion stimulates the JNK and p38 MAPK signaling transduction pathways, dissociation of ASK1-thioredoxin, and activation of ASK1. | Yu Y et al |
| 19255142 | 2009 | Opposing roles for ATF2 and c-Fos in c-Jun-mediated neuronal apoptosis. | Yuan Z et al |
| 11402340 | 2001 | Distinct roles of Jun : Fos and Jun : ATF dimers in oncogenesis. | van Dam H et al |
Other Information
Locus ID:
NCBI: 1386
MIM: 123811
HGNC: 784
Ensembl: ENSG00000115966
Variants:
dbSNP: 1386
ClinVar: 1386
TCGA: ENSG00000115966
COSMIC: ATF2
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA283 | MAPK8 | Gene | Pathway | associated | 23922006 | ||
| PA30621 | MAPK14 | Gene | Pathway | associated | 23922006 | ||
| PA445210 | Pain, Postoperative | Disease | ClinicalAnnotation | associated | PD | 30106255 | |
| PA449599 | fentanyl | Chemical | ClinicalAnnotation | associated | PD | 30106255 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38104370 | 2024 | Human umbilical cord mesenchymal stem cell-derived exosomes loaded miR-451a targets ATF2 to improve rheumatoid arthritis. | 4 |
| 38383842 | 2024 | MAP4K4 and WT1 mediate SOX6-induced cellular senescence by synergistically activating the ATF2-TGFβ2-Smad2/3 signaling pathway in cervical cancer. | 0 |
| 38104370 | 2024 | Human umbilical cord mesenchymal stem cell-derived exosomes loaded miR-451a targets ATF2 to improve rheumatoid arthritis. | 4 |
| 38383842 | 2024 | MAP4K4 and WT1 mediate SOX6-induced cellular senescence by synergistically activating the ATF2-TGFβ2-Smad2/3 signaling pathway in cervical cancer. | 0 |
| 36473315 | 2023 | Increased ATF2 expression predicts poor prognosis and inhibits sorafenib-induced ferroptosis in gastric cancer. | 16 |
| 36765266 | 2023 | Activating transcription factor-2 supports the antioxidant capacity and ability of human mesenchymal stem cells to prevent asthmatic airway inflammation. | 3 |
| 36842626 | 2023 | Analysis of the interaction of a non-canonical twin half-site of Cyclic AMP-Response Element (CRE) with CRE-binding protein. | 1 |
| 36939972 | 2023 | USP14 Regulates ATF2/PIK3CD Axis to Promote Microvascular Endothelial Cell Proliferation, Migration, and Angiogenesis in Diabetic Retinopathy. | 0 |
| 37237279 | 2023 | ATF2 loss promotes 5-FU resistance in colon cancer cells via activation of the ATR-Chk1 damage response pathway. | 0 |
| 37403209 | 2023 | ATF2 orchestrates macrophage differentiation and activation to promote antibacterial responses. | 0 |
| 37421203 | 2023 | The transcription factor ATF2 promotes gastric cancer progression by activating the METTL3/cyclin D1 pathway. | 0 |
| 37816820 | 2023 | The ATF2/miR-3913-5p/CREB5 axis is involved in the cell proliferation and metastasis of colorectal cancer. | 0 |
| 36473315 | 2023 | Increased ATF2 expression predicts poor prognosis and inhibits sorafenib-induced ferroptosis in gastric cancer. | 16 |
| 36765266 | 2023 | Activating transcription factor-2 supports the antioxidant capacity and ability of human mesenchymal stem cells to prevent asthmatic airway inflammation. | 3 |
| 36842626 | 2023 | Analysis of the interaction of a non-canonical twin half-site of Cyclic AMP-Response Element (CRE) with CRE-binding protein. | 1 |
Citation
Jean-Loup Huret
ATF2 (activating transcription factor 2)
Atlas Genet Cytogenet Oncol Haematol. 2012-10-01
Online version: http://atlasgeneticsoncology.org/gene/718/atf2
Historical Card
2007-07-01 ATF2 (activating transcription factor 2) by Pedro A Lazo,Ana Sevilla Affiliation
